KEYTRUDA® for neoadjuvant and adjuvant treatment of high-risk early-stage TNBC
-
EXPLORE KEYTRUDA® in the KEYNOTE-522 trial for patients with newly diagnosed previously untreated high-risk early-stage TNBC.
KEYNOTE-522 trial: A randomized, double-blind, multicenter, placebo-controlled study of KEYTRUDA® for the neoadjuvant and adjuvant treatment of patients with early-stage TNBC
Trial overview
KEYNOTE-522 was a randomized (2:1), double-blind, multicenter, placebo-controlled study evaluating the efficacy and safety of KEYTRUDA® (pembrolizumab) in combination with chemotherapy (carboplatin and paclitaxel followed by doxorubicin and cyclophosphamide [AC] or epirubicin and cyclophosphamide [EC]) given as a neoadjuvant treatment and continued as monotherapy adjuvant treatment after surgery, in newly diagnosed, previously untreated, high-risk early-stage TNBC patients (N=1,174)

Eligibility Criteria
- Newly diagnosed, not previously treated high-risk, early-stage TNBC
- Tumour size >1 cm but ≤2 cm in diameter with nodal involvement or tumour size >2 cm in diameter regardless of nodal involvement
- Enrolled regardless of tumour PD-L1 expression
Primary Endpoints:
- Pathologic complete response (pCR), defined as absence of invasive cancer in the breast and lymph nodes (ypT0/Tis ypN0) and assessed by the blinded local pathologist at the time of definitive surgery
- Event-free survival (EFS), defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause
Secondary Endpoints:
- Overall survival (OS)
AUC=area under the curve; EFS=event-free survival; OS=overall survival; pCR=pathologic complete response; Q3W=every 3 weeks; TNBC=triple-negative breast cancer; ypT0/Tis ypN0 = absence of invasive cancer in the breast and lymph nodes patients.
a All study medications were administered intravenously.
b Treatment with KEYTRUDA® or placebo continued until completion of the treatment (17 cycles), disease progression that precludes definitive surgery, disease recurrence in the adjuvant phase, or unacceptable toxicity.
c pCR (ypT0/Tis ypN0) was defined as absence of invasive cancer in the breast and lymph nodes and was assessed by the blinded local pathologist at the time of definitive surgery.
d EFS was defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause.
e At the time of EFS analysis, OS results were not yet mature (45% of the required events for final analysis).§ Carboplatin and paclitaxel followed by (doxorubicin or epirubicin) and cyclophosphamide.
* Based on a pre-specified EFS interim analysis (compared to a significance level of 0.0052).
‡ Based on Miettinen and Nurminen method stratified by nodal status, tumour size, and choice of carboplatin.
† Based on a follow-up analysis in the entire intention-to-treat population (n=1,174), the pCR rate difference was 7.5 (95% CI: 1.6, 13.4).
¶ Based on Cox regression model with Efron’s method of tie handling with treatment as a covariate stratified by nodal status, tumour size, and choice of carboplatin.
# Based on log-rank test stratified by nodal status, tumour size, and choice of carboplatin.AUC=area under the curve; EFS=event-free survival; OS=overall survival; pCR=pathologic complete response; Q3W=every 3 weeks; TNBC=triple-negative breast cancer; ypT0/Tis ypN0 = absence of invasive cancer in the breast and lymph nodes patients.
a All study medications were administered intravenously.
b Treatment with KEYTRUDA® or placebo continued until completion of the treatment (17 cycles), disease progression that precludes definitive surgery, disease recurrence in the adjuvant phase, or unacceptable toxicity.
c pCR (ypT0/Tis ypN0) was defined as absence of invasive cancer in the breast and lymph nodes and was assessed by the blinded local pathologist at the time of definitive surgery.
d EFS was defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause.
e At the time of EFS analysis, OS results were not yet mature (45% of the required events for final analysis).§ Carboplatin and paclitaxel followed by (doxorubicin or epirubicin) and cyclophosphamide.
* Based on a pre-specified EFS interim analysis (compared to a significance level of 0.0052).
‡ Based on Miettinen and Nurminen method stratified by nodal status, tumour size, and choice of carboplatin.
† Based on a follow-up analysis in the entire intention-to-treat population (n=1,174), the pCR rate difference was 7.5 (95% CI: 1.6, 13.4).
¶ Based on Cox regression model with Efron’s method of tie handling with treatment as a covariate stratified by nodal status, tumour size, and choice of carboplatin.
# Based on log-rank test stratified by nodal status, tumour size, and choice of carboplatin.Pathological Complete Response
Neoadjuvant KEYTRUDA® + chemotherapy/adjuvant KEYTRUDA® demonstrated a statistically significant improvement in pCR vs. placebo + chemotherapy/placebo
13.6% more patients had a pCR with KEYTRUDA® (pembrolizumab) + chemotherapy regimen§ vs placebo + chemotherapy regimen§ (95% CI, 5.4–21.8; p=0.00055)*‡
KEYTRUDA® + chemotherapy/KEYTRUDA® (n=401/602)Placebo + chemotherapy/placebo (n=201/602)Number of patients with pCR (ypT0/Tis ypN0)*†260103pCR rate (%) (ypT0/Tis ypN0)*64.8 (95% CI: 59.9, 69.5)51.2 (95% CI: 44.1, 58.3)Treatment difference estimate (%)†‡13.6 (95% CI: 5.4, 21.8), p=0.00055Event-Free Survival
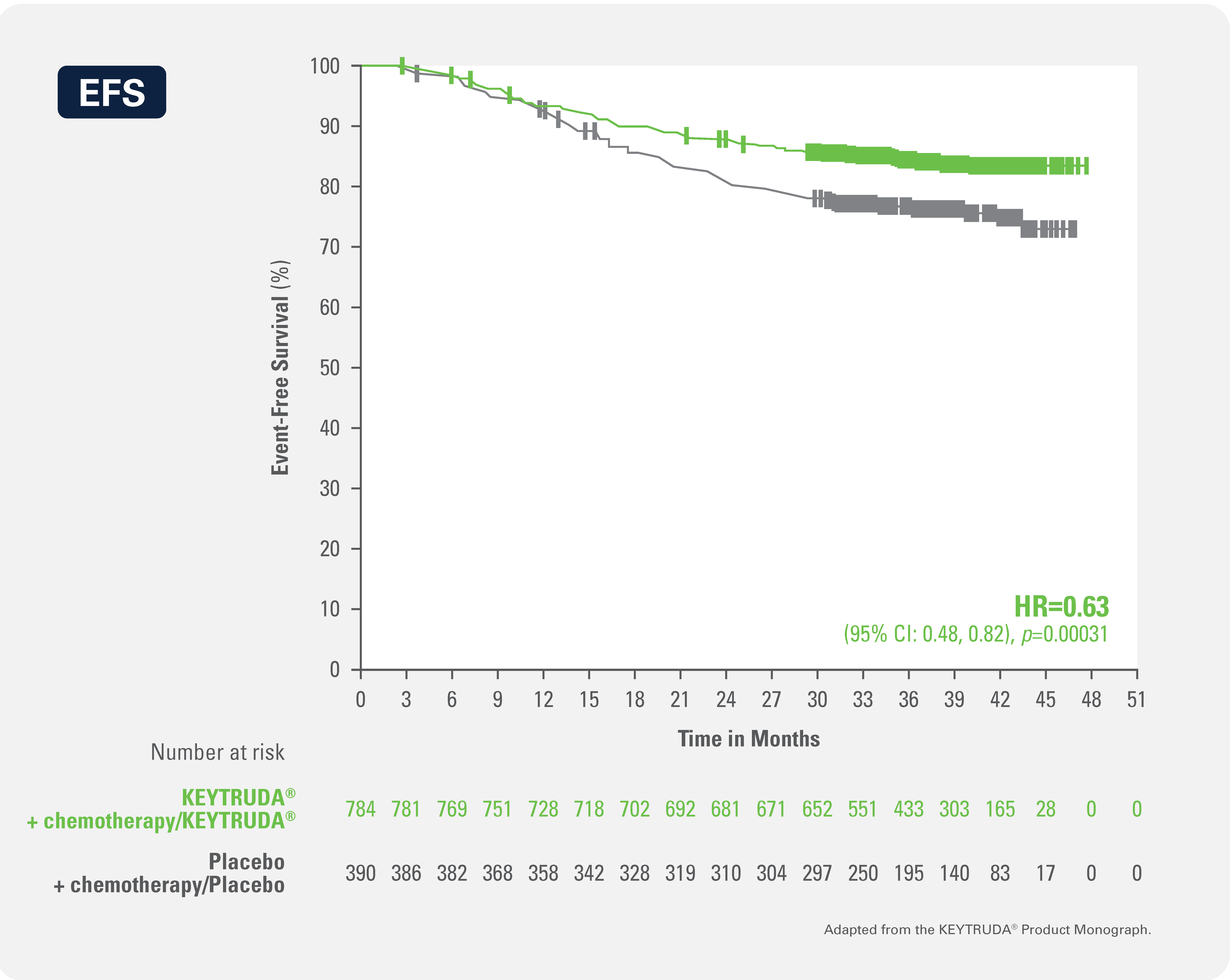
Neoadjuvant KEYTRUDA® + chemotherapy/adjuvant KEYTRUDA® demonstrated a statistically significant improvement in EFS* (HR: 0.63 [95% CI: 0.48, 0.82]¶, p=0.00031#) vs. placebo + chemotherapy/placebo and a 37% reduction in instantaneous risk of events
Kaplan-Meier Curve for Event-Free Survival by Treatment Arm in KEYNOTE-522 (Intent to Treat Population)
Overall Survival
At the time of EFS analysis, OS results were not yet mature (45% of the required events for final analysis).
AUC=area under the curve; EFS=event-free survival; OS=overall survival; pCR=pathologic complete response; Q3W=every 3 weeks; TNBC=triple-negative breast cancer; ypT0/Tis ypN0 = absence of invasive cancer in the breast and lymph nodes patients.
a All study medications were administered intravenously.
b Treatment with KEYTRUDA® or placebo continued until completion of the treatment (17 cycles), disease progression that precludes definitive surgery, disease recurrence in the adjuvant phase, or unacceptable toxicity.
c pCR (ypT0/Tis ypN0) was defined as absence of invasive cancer in the breast and lymph nodes and was assessed by the blinded local pathologist at the time of definitive surgery.
d EFS was defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause.
e At the time of EFS analysis, OS results were not yet mature (45% of the required events for final analysis).
§ Carboplatin and paclitaxel followed by (doxorubicin or epirubicin) and cyclophosphamide.
* Based on a pre-specified EFS interim analysis (compared to a significance level of 0.0052).
‡ Based on Miettinen and Nurminen method stratified by nodal status, tumour size, and choice of carboplatin.
† Based on a follow-up analysis in the entire intention-to-treat population (n=1,174), the pCR rate difference was 7.5 (95% CI: 1.6, 13.4).
¶ Based on Cox regression model with Efron’s method of tie handling with treatment as a covariate stratified by nodal status, tumour size, and choice of carboplatin.
# Based on log-rank test stratified by nodal status, tumour size, and choice of carboplatin.
AUC=area under the curve; EFS=event-free survival; OS=overall survival; pCR=pathologic complete response; Q3W=every 3 weeks; TNBC=triple-negative breast cancer; ypT0/Tis ypN0 = absence of invasive cancer in the breast and lymph nodes patients.
a All study medications were administered intravenously.
b Treatment with KEYTRUDA® or placebo continued until completion of the treatment (17 cycles), disease progression that precludes definitive surgery, disease recurrence in the adjuvant phase, or unacceptable toxicity.
c pCR (ypT0/Tis ypN0) was defined as absence of invasive cancer in the breast and lymph nodes and was assessed by the blinded local pathologist at the time of definitive surgery.
d EFS was defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause.
e At the time of EFS analysis, OS results were not yet mature (45% of the required events for final analysis).§ Carboplatin and paclitaxel followed by (doxorubicin or epirubicin) and cyclophosphamide.
* Based on a pre-specified EFS interim analysis (compared to a significance level of 0.0052).
‡ Based on Miettinen and Nurminen method stratified by nodal status, tumour size, and choice of carboplatin.
† Based on a follow-up analysis in the entire intention-to-treat population (n=1,174), the pCR rate difference was 7.5 (95% CI: 1.6, 13.4).
¶ Based on Cox regression model with Efron’s method of tie handling with treatment as a covariate stratified by nodal status, tumour size, and choice of carboplatin.
# Based on log-rank test stratified by nodal status, tumour size, and choice of carboplatin.KEYTRUDA® established safety profile in high-risk early-stage TNBC
Most common Grades 3 to 5 treatment-related adverse events (reported in ≥ 5% of patients) were:
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionKEYTRUDA® 200 mg every 3 weeks with Chemotherapy*
/KEYTRUDA® 200 mg every 3 weeks
(n=783)Placebo with Chemotherapy*
/Placebo
(n=389)Neutropenia34.5%33.4%Neutrophil Count Decreased18.6%23.1%Anemia18%14.9%Febrile Neutropenia17.8%16%White blood cell count decreased7.7%5.2%Alanine aminotransferase increased5.5%2.3%- Fatal adverse events regardless of causality to the study treatment occurred in 0.9% of patients receiving KEYTRUDA® in combination with chemotherapy as neoadjuvant treatment, and then continued as monotherapy as adjuvant treatment after surgery, including 1 each of autoimmune encephalitis, pneumonia, pneumonitis, pulmonary embolism, sepsis in association with multiple organ dysfunction syndrome and myocardial infarction, shock, and death from unknown cause
- Serious treatment-related adverse events occurred in 34% of patients receiving KEYTRUDA®
- KEYTRUDA® was interrupted for treatment-related adverse events in 49% of patients
Treatment-related adverse events, any grade (reported in ≥ 20% of patients) were:
Adapted from the KEYTRUDA® Product Monograph.Adverse EventKEYTRUDA® 200 mg every 3 weeks with Chemotherapy*
/KEYTRUDA® 200 mg every 3 weeks
(n=783)Placebo with Chemotherapy*
/Placebo
(n=389)Nausea63.2%63.0%Alopecia60.2%56.6%Anemia54.8%55.3%Neutropenia46.9%47.6%Fatigue42.1%38.8%Diarrhea30.4%25.2%Alanine aminotransferase increased26.1%25.2%Neuropathy peripheral19.7%21.6%Decreased appetite19.5%14.7%AUC=area under the curve; EFS=event-free survival; OS=overall survival; pCR=pathologic complete response; Q3W=every 3 weeks; TNBC=triple-negative breast cancer; ypT0/Tis ypN0 = absence of invasive cancer in the breast and lymph nodes patients.
a All study medications were administered intravenously.
b Treatment with KEYTRUDA® or placebo continued until completion of the treatment (17 cycles), disease progression that precludes definitive surgery, disease recurrence in the adjuvant phase, or unacceptable toxicity.
c pCR (ypT0/Tis ypN0) was defined as absence of invasive cancer in the breast and lymph nodes and was assessed by the blinded local pathologist at the time of definitive surgery.
d EFS was defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause.
e At the time of EFS analysis, OS results were not yet mature (45% of the required events for final analysis).§ Carboplatin and paclitaxel followed by (doxorubicin or epirubicin) and cyclophosphamide.
* Based on a pre-specified EFS interim analysis (compared to a significance level of 0.0052).
‡ Based on Miettinen and Nurminen method stratified by nodal status, tumour size, and choice of carboplatin.
† Based on a follow-up analysis in the entire intention-to-treat population (n=1,174), the pCR rate difference was 7.5 (95% CI: 1.6, 13.4).
¶ Based on Cox regression model with Efron’s method of tie handling with treatment as a covariate stratified by nodal status, tumour size, and choice of carboplatin.
# Based on log-rank test stratified by nodal status, tumour size, and choice of carboplatin.
KEYTRUDA® in the first-line setting for locally recurrent unresectable or metastatic TNBC
-
EXPLORE KEYTRUDA® in the KEYNOTE-355 trial for patients with locally recurrent unresectable or metastatic TNBC, which had not been previously treated with chemotherapy in the metastatic setting.
KEYNOTE-355 trial: A randomized, double-blind, multicenter, placebo controlled study of KEYTRUDA® for the treatment of locally recurrent unresectable or metastatic TNBC
Trial overview
Keynote-355 was a randomized, double-blind trial of KEYTRUDA® + chemotherapy vs. placebo + chemotherapy in treatment-naïve patients with locally recurrent unresectable or metastatic TNBC regardless of tumour PD-L1 expression¶‖. A total of 847 patients were randomized (2:1) and patients received either KEYTRUDA® 200 mg IV Q3W (n=556) or placebo IV Q3W (n=281).
KEYTRUDA® is indicated for treatment in combination with chemotherapy for adult patients with locally recurrent unresectable or metastatic triple-negative breast cancer (TNBC), who have not received prior chemotherapy for metastatic disease and whose tumours express PD-L1 ( [CPS] ≥ 10) as determined by a validated test.
Treatment Arms
KEYTRUDA® 200 mg on Day 1 every 3 weeks in combination with
nab-paclitaxel 100 mg/m2 on Days 1, 8 and 15 every 28 days, or
paclitaxel 90 mg/m2 on Days 1, 8, and 15 every 28 days, or
gemcitabine 1000 mg/m2 and carboplatin AUC 2 mg/mL/min on Days 1 and 8 every 21 days
(n=566; CPS ≥ 10: n=220)
Placebo on Day 1 every 3 weeks in combination with
nab-paclitaxel 100 mg/m2 on Days 1, 8 and 15 every 28 days, or
paclitaxel 90 mg/m2 on Days 1, 8, and 15 every 28 days, or
gemcitabine 1000 mg/m2 and carboplatin AUC 2 mg/mL/min on Days 1 and 8 every 21 days
(n=281; CPS ≥ 10: n=103)
Key Eligibility Criteria
- Age ≥18 years
- Centrally confirmed TNBC (ASCO-CAP guideline criteria)
- ≥1 measurable lesion on RECIST v1.1
- Tumour sample from a locally recurrent inoperable or metastatic site
- Completion of treatment with curative intent ≥6 months before first disease recurrence
- ECOG PS 0 or 1
- Adequate organ function
Major Efficacy Outcomes:
- Progression-free survival based on RECIST version 1.1 as assessed by BICR, modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ with tumour PD-L1 expression CPS ≥ 10
- Overall survival in patients with tumour PD-L1 expression CPS ≥ 10
Additional Efficacy Outcomes:
- Objective response rate in patients with tumour PD-L1 expression CPS ≥10
- Duration of response in patients with tumour PD-L1 expression CPS ≥10
ASCO-CAP=American Society of Clinical Oncology-College of American Pathologists; BICR=Blinded Independent Central Review; CI=confidence intervalsCPS=combined positive score; ECOG PS=Eastern Cooperative Oncology Group performance status; HR=hazard ratio; TNBC=triple-negative breast cancer; PD-L1=programmed death ligand-1; Q3W=every 3 weeks; RECIST=Response Evaluation Criteria In Solid Tumours.
¶ 38% of the total 847 patients had tumor PD-L1 expression CPS ≥ 10
‡ Based on stratified Cox regression model.
Þ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.00411).
§ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.0113).
*Chemotherapy: paclitaxel, nab-paclitaxel, or gemcitabine and carboplatin.
# Based on the pre-specified final analysis (data cutoff – 15 June 2021).
† Based on RECIST version 1.1 as assessed by BICR.
** Based on a pre-specified interim analysis (data cutoff – 11 December 2019).ASCO-CAP=American Society of Clinical Oncology-College of American Pathologists; BICR=Blinded Independent Central Review; CI=confidence intervalsCPS=combined positive score; ECOG PS=Eastern Cooperative Oncology Group performance status; HR=hazard ratio; TNBC=triple-negative breast cancer; PD-L1=programmed death ligand-1; Q3W=every 3 weeks; RECIST=Response Evaluation Criteria In Solid Tumours.
¶ 38% of the total 847 patients had tumor PD-L1 expression CPS ≥ 10
‡ Based on stratified Cox regression model.
Þ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.00411).
§ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.0113).
*Chemotherapy: paclitaxel, nab-paclitaxel, or gemcitabine and carboplatin.
# Based on the pre-specified final analysis (data cutoff – 15 June 2021).
† Based on RECIST version 1.1 as assessed by BICR.
** Based on a pre-specified interim analysis (data cutoff – 11 December 2019).Progression Free Survival
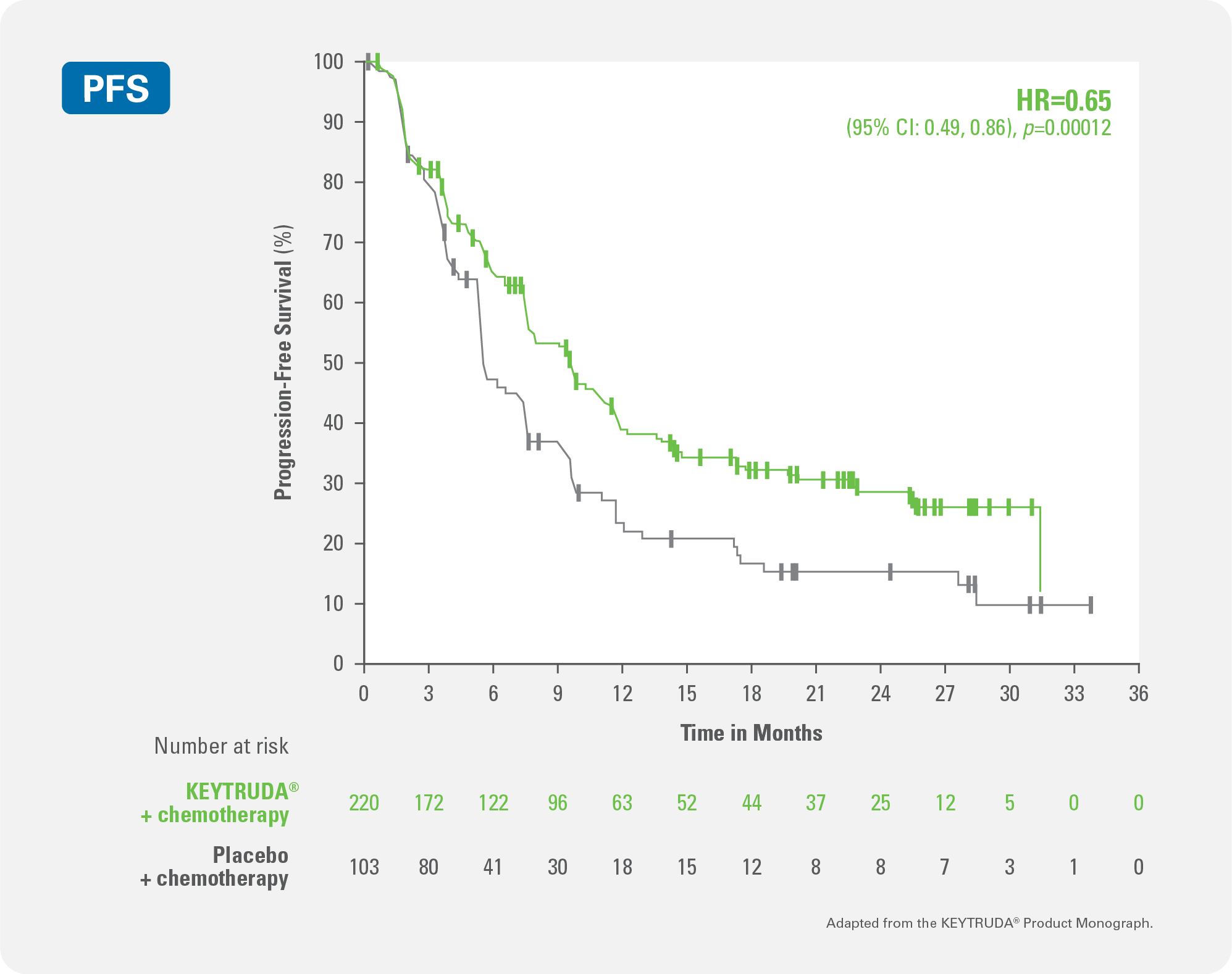
KEYTRUDA® + chemotherapy demonstrated significant improvement in progression-free survival (PFS) vs. placebo + chemotherapy
35% reduction in instantaneous risk of death or disease progression for KEYTRUDA® + chemotherapy vs. placebo + chemotherapy (HR‡=0.65; 95% CI:0.49, 0.86; pÞ,§=0.0012)
Kaplan-Meier Curve for Progression Free Survival by Treatment Arm in KEYNOTE-355 (CPS ≥ 10)
Median Progression-Free-Survival
Median Progression-Free Survival for patients with locally recurrent unresectable or metastatic TNBC with PD-L1 expression CPS ≥ 10
mPFS in monthsKEYTRUDA® + chemotherapy*
(n=220)Placebo + chemotherapy*
(n=103)9.7 months
(95% CI: 7.6, 11.3)5.6 months
(95% CI: 5.3, 7.5)Overall Survival
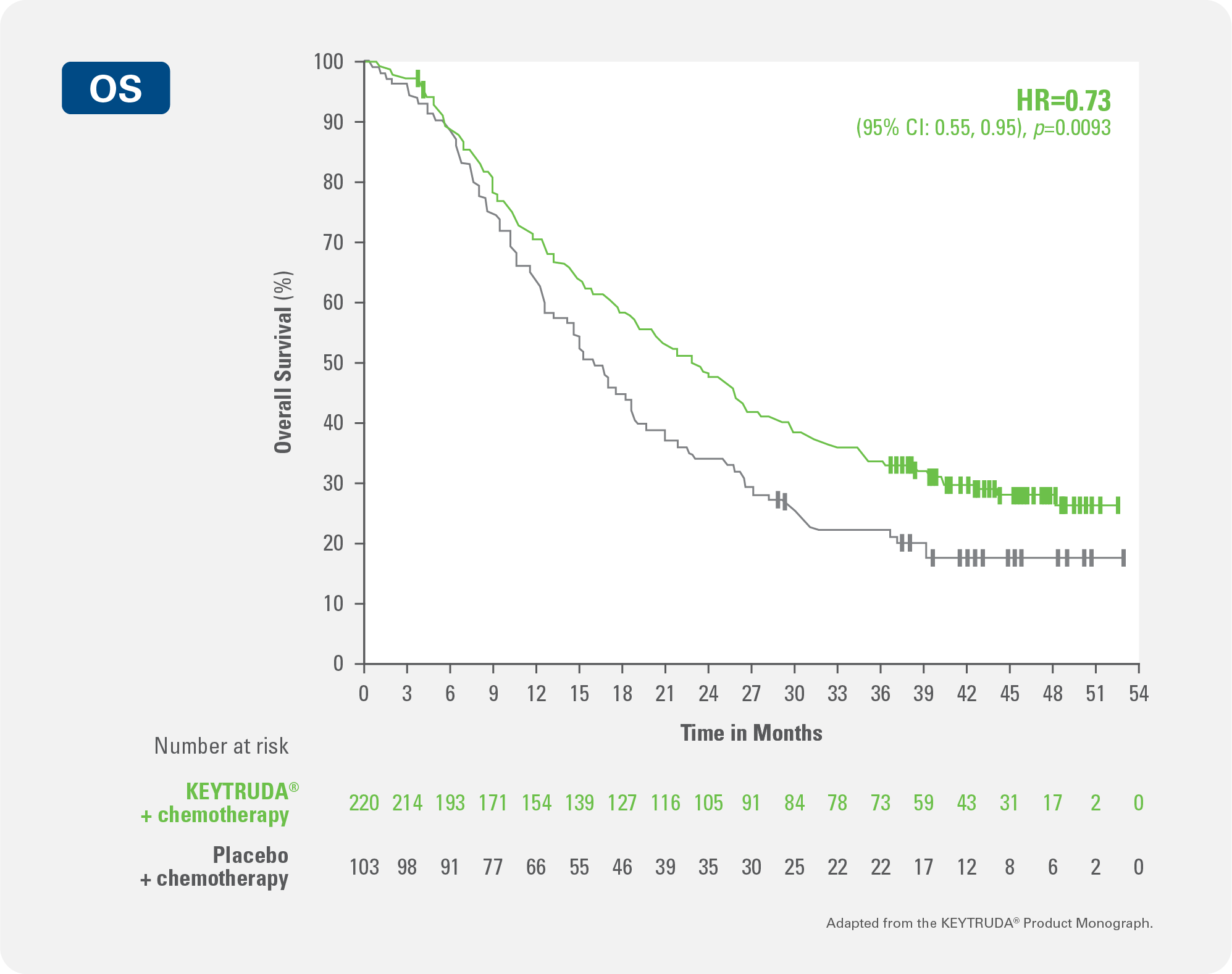
KEYTRUDA® + chemotherapy demonstrated significant improvement in overall survival (OS) vs. placebo + chemotherapy
27% reduction in instantaneous risk of death or disease progression for KEYTRUDA® + chemotherapy vs. placebo + chemotherapy (HR#=0.73 (0.55, 0.95); p§=0.0093)
Kaplan-Meier Curve for Overall Survival by Treatment Arm in KEYNOTE-355 (CPS ≥10)
Objective Response Rate
Objective Response Rate for patients with locally recurrent unresectable or metastatic TNBC with PD-L1 expression CPS ≥ 10
ORR, (95% CI)KEYTRUDA® + chemotherapy*
(n=220)Placebo + chemotherapy*
(n=103)53% (95% CI: 46, 59)41% (95% CI: 31, 51)Duration of Response
Duration of response, median in months (range) of patients with locally recurrent unresectable or metastatic TNBC with PD-L1 expression CPS ≥ 10 †**
KEYTRUDA® + chemotherapy*
(n=220)Placebo + chemotherapy*
(n=103)12.87.3ASCO-CAP=American Society of Clinical Oncology-College of American Pathologists; BICR=Blinded Independent Central Review; CI=confidence intervalsCPS=combined positive score; ECOG PS=Eastern Cooperative Oncology Group performance status; HR=hazard ratio; TNBC=triple-negative breast cancer; PD-L1=programmed death ligand-1; Q3W=every 3 weeks; RECIST=Response Evaluation Criteria In Solid Tumours.
¶ 38% of the total 847 patients had tumor PD-L1 expression CPS ≥ 10
‡ Based on stratified Cox regression model.
Þ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.00411).
§ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.0113).
*Chemotherapy: paclitaxel, nab-paclitaxel, or gemcitabine and carboplatin.
# Based on the pre-specified final analysis (data cutoff – 15 June 2021).
† Based on RECIST version 1.1 as assessed by BICR.
** Based on a pre-specified interim analysis (data cutoff – 11 December 2019).ASCO-CAP=American Society of Clinical Oncology-College of American Pathologists; BICR=Blinded Independent Central Review; CI=confidence intervalsCPS=combined positive score; ECOG PS=Eastern Cooperative Oncology Group performance status; HR=hazard ratio; TNBC=triple-negative breast cancer; PD-L1=programmed death ligand-1; Q3W=every 3 weeks; RECIST=Response Evaluation Criteria In Solid Tumours.
¶ 38% of the total 847 patients had tumor PD-L1 expression CPS ≥ 10
‡ Based on stratified Cox regression model.
Þ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.00411).
§ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.0113).
*Chemotherapy: paclitaxel, nab-paclitaxel, or gemcitabine and carboplatin.
# Based on the pre-specified final analysis (data cutoff – 15 June 2021).
† Based on RECIST version 1.1 as assessed by BICR.
** Based on a pre-specified interim analysis (data cutoff – 11 December 2019).Demonstrated safety profile in locally recurrent unresectable or metastatic TNBC
The most common Grade 3-5 treatment-related adverse events (reported in ≥20% of patients) for patients treated with KEYTRUDA®
Adapted from the KEYTRUDA® Product Monograph.Adverse eventKEYTRUDA® + chemotherapy (n=596)Placebo + chemotherapy (n=281)Neutropenia29.229.9Neutrophil count decreased17.311.0Anemia16.414.6White blood cell count decreased10.210.3Thrombocytopenia9.911.1Leukopenia9.710.7Platelet count decreased6.04.3Alanine aminotransferase increased5.74.6- KEYTRUDA® was discontinued for treatment-related adverse events in 9.1% of patients
- Fatal treatment-related adverse-events occurred in 0.3% of patients receiving KEYTRUDA® in combination with chemotherapy including 1 case each of pneumonia and acute kidney injury
- Serious treatment-related adverse events occurred in 17.6% of patients receiving KEYTRUDA® in combination with chemotherapy
- KEYTRUDA® was interrupted for treatment-related adverse events in 43% of patients
Most common treatment-related adverse events (reported in ≥20% of patients), any grade with either KEYTRUDA® + chemotherapy or placebo + chemotherapy
Adapted from the KEYTRUDA® Product Monograph.Adverse eventKEYTRUDA® + chemotherapy (n=596)Placebo + chemotherapy (n=281)Anemia48.845.9Neutropenia40.438.1Nausea38.440.9Alopecia33.133.5Fatigue27.529.5Neutrophil count decreased22.126.3ASCO-CAP=American Society of Clinical Oncology-College of American Pathologists; BICR=Blinded Independent Central Review; CI=confidence intervalsCPS=combined positive score; ECOG PS=Eastern Cooperative Oncology Group performance status; HR=hazard ratio; TNBC=triple-negative breast cancer; PD-L1=programmed death ligand-1; Q3W=every 3 weeks; RECIST=Response Evaluation Criteria In Solid Tumours.
¶ 38% of the total 847 patients had tumor PD-L1 expression CPS ≥ 10
‡ Based on stratified Cox regression model.
Þ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.00411).
§ One-sided p-Value based on stratified log-rank test (compared to a significance level of 0.0113).
*Chemotherapy: paclitaxel, nab-paclitaxel, or gemcitabine and carboplatin.
# Based on the pre-specified final analysis (data cutoff – 15 June 2021).
† Based on RECIST version 1.1 as assessed by BICR.
** Based on a pre-specified interim analysis (data cutoff – 11 December 2019).
References
1. KEYTRUDA® Product Monograph. Merck Canada Inc. February 6, 2024.
2. Schmid et al. Event-free Survival with Pembrolizumab in Early Triple-Negative Breast Cancer. N Eng J Medicine. 2022:386(6);556-567.
CA-OBR-00005

