KEYTRUDA® as adjuvant therapy in resected stage III melanoma1
-
KEYTRUDA® in the KEYNOTE-054 trial was studied in patients with resected stage III melanoma with lymph nodes involvement
KEYNOTE-054 trial: Controlled phase 3 trial of KEYTRUDA® in the adjuvant treatment of patients with completely resected stage III (A, B, or C) melanoma
Trial overview
- Multicentre, randomized double-blind, placebo-controlled trial
- 1019 patients with completely resected stage IIIA (>1 mm lymph node metastasis),
IIIB or IIIC melanoma - Patients must have undergone lymph node dissection and if indicated, radiotherapy
within 13 weeks prior to starting treatment - The study design included reinitiation with KEYTRUDA® for subsequent disease
recurrence that occurred >6 months after completion of one year of adjuvant treatment
Treatment arms
KEYTRUDA® 200 mg Q3W (n=514)
Placebo (n=505)
Primary endpoints
- Investigator-assessed:
- Recurrence free survival in the intent-to-treat population
- Recurrence free survival in the subgroup of patients with PD-L1 positive tumours
- Recurrence-free survival was defined as the time between the date of randomization and the date of first recurrence (local, regional, or distant metastasis) or death, whichever occurs first
Secondary endpoints
- Distant metastasis-free survival (DMFS) in the ITT population and in the subgroup of patients with PD-L1 positive tumours
- Overall survival (OS) in the ITT population and in the subgroup of patients with PD-L1 positive tumours
AE= adverse events; CI=confidence interval; HR=hazard ratio; DMFS=distant metastasis-free survival; NR=not reached; PD-L1=programmed death-ligand 1; Q3W=every three weeks; RFS= recurrence-free survival
* At pre-specified interim analysis
† Based on the stratified Cox proportional hazard model
‡ p-Value (based on stratified log rank test) is compared with 0.014 of the allocated alpha for this interim analysis.
§ At DMFS analysis.ITT population (interim analysis)
Recurrence Free Survival
KEYTRUDA® demonstrated significant improvements in recurrence-free survival at pre-specified analysis (primary endpoint).
43% reduction in the risk of recurrence or death* HR=0.57† (98% CI: 0.43, 0.74; p<0.0001‡)
KEYTRUDA® 200 mg every 3 weeks
n=514Placebo
n=505RFS26%43%HR=0.57† (98% CI: 0.43, 0.74; p<0.0001‡)Kaplan-Meier Curve for Recurrence-Free Survival in the KEYNOTE-054 trial (Intent to Treat Population)

Distant Metastasis-free Survival
KEYTRUDA® demonstrated significant improvements in distant metastasis-free survival (secondary endpoint).
40% reduction in the risk of distant metastasis§ HR=0.60†(95% CI: 0.49, 0.73; p<0.0001‡)
KEYTRUDA® 200 mg every 3 weeks
n=514Placebo
n=505DMFS34%49%HR=0.60†(95% CI: 0.49, 0.73; p<0.0001‡)Patients with PD-L1 positive tumors
Recurrence free and distant free-metastasis in a subpopulation of patients with PD-L positive tumors
RFS for KEYTRUDA® vs placebo
(n=427)(primary endpoint)HR=0.54 (95% CI: 0.42, 0.69)DMFS for KEYTRUDA® vs placebo
(n=427)(secondary endpoint)HR=0.61 (95% CI: 0.49, 0.76)Results at the time of the analysis, overall survival had not been formally assessed.
AE= adverse events; CI=confidence interval; HR=hazard ratio; DMFS=distant metastasis-free survival; NR=not reached; PD-L1=programmed death-ligand 1; Q3W=every three weeks; RFS= recurrence-free survival
* At pre-specified interim analysis
† Based on the stratified Cox proportional hazard model
‡ p-Value (based on stratified log rank test) is compared with 0.014 of the allocated alpha for this interim analysis.
§ At DMFS analysis.KEYTRUDA® demonstrated safety profile results in melanoma in the KEYNOTE-054 trial
The most common grades 3 to 4 treatment-related adverse events (reported in ≥1% of patients) were:
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionKEYTRUDA® 200 mg
every 3 weeks
% (n=509)Placebo
% (n=502)Colitis1.20.2Type 1 diabetes melitus1.00Lipase increased0.80.2- Treatment-related adverse events leading to discontinuation of KEYTRUDA® occurred in 12% of patients
- The most common treatment-related adverse event resulting in discontinuation of KEYTRUDA® was pneumonitis (n=7, 1.4%)
- There were 2 (0.4%) deaths reported in the KEYTRUDA® arm: drug reaction with eosinophilia and systemic symptoms (n=1); and autoimmune myositis with respiratory failure (n=1).
- The median time to discontinuation for treatment-related adverse-events was 5.8 months.
Treatment-related adverse events, any grade (reported in ≥10% of patients) were:
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionKEYTRUDA® 200 mg
every 3 weeks
% (n=509)Placebo
% (n=502)Fatigue28.126.9Diarrhea18.516.3Pruritus16.79.8Hypothyroidism14.32.6Nausea11.48.6Arthralgia10.09.4For the complete list of treatment-related adverse events, please consult the Product Monograph
AE= adverse events; CI=confidence interval; HR=hazard ratio; DMFS=distant metastasis-free survival; NR=not reached; PD-L1=programmed death-ligand 1; Q3W=every three weeks; RFS= recurrence-free survival
* At pre-specified interim analysis
† Based on the stratified Cox proportional hazard model
‡ p-Value (based on stratified log rank test) is compared with 0.014 of the allocated alpha for this interim analysis.
§ At DMFS analysis.
KEYTRUDA® in the first-line setting for metastatic melanoma1
-
KEYTRUDA® in the KEYNOTE-006 trial was studied in patients with unresectable or metastatic melanoma
Keynote-006 trial: Controlled phase 3 trial of KEYTRUDA® in patients with unresectable or metastatic melanoma naïve to treatment with ipilimumab
Trial overview
- Randomized, multicenter, controlled trial
- 834 melanoma in patients who were naïve to ipilimumab and who received no prior systemic therapy
- Patients with BRAF V600E mutant melanoma were not required to have received prior BRAF inhibitor therapy
KEYTRUDA®10 mg/kg IV Q2W (n=279)
KEYTRUDA®10 mg/kg IV Q3W (n=277)
Ipilimumab 3 mg/kg IV Q3W (n=278)
KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
Primary endpoints
- Overall survival
- Progression-free survival, as assessed by IRO review using RECIST 1.1
Secondary endpoints
- Overall response rate, as assessed by IRO review using RECIST 1.1
- Response duration, as assessed by IRO review using RECIST 1.1
AE = adverse event; IRO= independent radiology plus oncologist; IV= intravenous; Q2W= every 2 weeks; Q3W= every 3 weeks; RECIST=response evaluation criteria in solid tumours; CI=confidence interval; HR=hazard ratio; IRO= independent radiology plus oncologist review using RECIST 1.1; OS=overall survival; PFS= progression-free survival;
* At prespecified interim analysis
† Based on the Cox proportional hazard model stratified by line of therapy, ECOG performance status, and PD-L1 expression status
‡ p-Value based on stratified log rank test
aBased on the final analysis with an additional follow-up of 9 months (total of 566 events)
§Based on patients with a best overall response as confirmed complete or partial response
bfor all patients as treatedOS and PFS data in patients with unresectable or metastatic melanoma naïve to treatment with ipilimumab.
ITT population (Intent-to-Treat Analysis)
Overall Survival
Interim AnalysisOSKEYTRUDA® 10 mg/kg Q2W vs IpilimumabHR=0.63†(95% CI: 0.47, 0.83; p=0.00052‡)KEYTRUDA® 10 mg/kg Q3W vs IpilimumabHR=0.69† (95% CI: 0.52, 0.90; p=0.00358 ‡)KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
Kaplan-Meier Curve for Overall Survival in the KEYNOTE-006 trial (Intent to Treat Population) (Final Analysis)

Based on the final analysis with an additional follow-up of 9 months (total of 383 deaths as pre-specified in the protocol
Progression-Free Survival
Interim AnalysisPFSKEYTRUDA® 10 mg/kg Q2W
vs Ipilimumab (n=279)HR=0.58†(95% CI: 0.47, 0.72; p<0.000011‡)KEYTRUDA® 10 mg/kg Q3W
vs Ipilimumab (n=277)HR=0.58†(95% CI: 0.47, 0.72; p<0.000011‡)KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
Kaplan-Meier Curve for Progression-free Survival in the KEYNOTE-006 trial (Intent to Treat Population) (Final analysis)

Based on the final analysis with an additional follow-up of 9 months (total of 566 events)
Overall Response Rate
Secondary outcomes measured by IRO§*
KEYTRUDA®
10 mg/kg Q2W n=279KEYTRUDA®
10 mg/kg Q3W n=277Ipilimumab
n=278ORR %
(95% CI)34% (28, 40)33% (27, 39)12% (8, 16)mDOR8.3 months
(1.4+, 8.3)Not reached
(1.4+, 8.1+)Not reached
(1.1+, 7.9+)KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
Efficacy results for a subpopulation analysis by PD-L1 status for pooled KEYTRUDA®
[10 mg/kg Q2W or Q3W] vs ipilimumabAmong patients who were evaluable for PD-L1 expression, 82% were positive and 18% were PD-L1 negative
PD-L1 positive
(n=447)PD-L1 negative
(n=98)OSHR=0.56
(95% CI: 0.43, 0.73)HR=0.95
(95% CI: 0.56, 1.62)PFSHR=0.53
(95% CI: 0.43, 0.65)HR=0.73
(95% CI: 0.47, 1.11)KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
Efficacy results for a subpopulation analysis by BRAF mutation status for pooled KEYTRUDA® [10 mg/kg Q2W or Q3W] vs ipilimumab
BRAF wild-typeBRAF mutant without prior BRAF treatmentBRAF mutant with prior BRAF treatmentOSHR=0.61
(95% CI: 0.46, 0.82)HR=0.69
(95% CI: 0.33, 1.45)HR=0.75 (95% CI: 0.45, 1.26)PFSHR=0.57
(95% CI: 0.45, 0.73)HR=0.50
(95% CI: 0.32, 0.77)HR=0.73
(95% CI: 0.48, 1.11)KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
AE = adverse event; IRO= independent radiology plus oncologist; IV= intravenous; Q2W= every 2 weeks; Q3W= every 3 weeks; RECIST=response evaluation criteria in solid tumours; CI=confidence interval; HR=hazard ratio; IRO= independent radiology plus oncologist review using RECIST 1.1; OS=overall survival; PFS= progression-free survival;
* At prespecified interim analysis
† Based on the Cox proportional hazard model stratified by line of therapy, ECOG performance status, and PD-L1 expression status
‡ p-Value based on stratified log rank test
aBased on the final analysis with an additional follow-up of 9 months (total of 566 events)
§Based on patients with a best overall response as confirmed complete or partial response
bfor all patients as treatedKEYTRUDA® safety profile results in unresectable or metastatic melanoma, in the KEYNOTE-006 trial
The most common grades 3-4 treatment-related adverse events wereb:
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionsKEYTRUDA® 10 mg/kg every 2 or 3 weeks
% (n=555)Ipilimumab 3 mg/kg Q3W
% (n=256)Diarrhea1.83.1Colitis1.76.3Fatigue0.21.2KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
- Adverse reactions leading to permanent discontinuation of KEYTRUDA® occurred in 9% of patients.
- Adverse reactions leading to interruption of KEYTRUDA® occurred in 21% of patients.
- The most common adverse reactions (reported in at least 20% of patients) were fatigue and diarrhea.
Treatment-related adverse events, any grade (reported in ≥10% of patients) wereb:
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionsKEYTRUDA® 10 mg/kg every 2 or 3 weeks
% (n=555)Ipilimumab 3 mg/kg Q3W
% (n=256)Fatigue2015.2Diarrhea15.722.7Pruritus14.225.4Rash14.114.5Asthenia11.46.3Nausea10.68.6Arthralgia10.55.1Vitiligo10.11.6KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
For the complete list of treatment-related adverse events, please consult the Product Monograph.
AE = adverse event; IRO= independent radiology plus oncologist; IV= intravenous; Q2W= every 2 weeks; Q3W= every 3 weeks; RECIST=response evaluation criteria in solid tumours; CI=confidence interval; HR=hazard ratio; IRO= independent radiology plus oncologist review using RECIST 1.1; OS=overall survival; PFS= progression-free survival;
* At prespecified interim analysis
† Based on the Cox proportional hazard model stratified by line of therapy, ECOG performance status, and PD-L1 expression status
‡ p-Value based on stratified log rank test
aBased on the final analysis with an additional follow-up of 9 months (total of 566 events)
§Based on patients with a best overall response as confirmed complete or partial response
bfor all patients as treated
KEYTRUDA® in the second-line setting for metastatic melanoma1
-
KEYTRUDA® in the KEYNOTE-002 trial was studied patients with unresectable or metastatic melanoma
Keynote-002 trial: Controlled phase II trial in patients with unresectable or metastatic melanoma previously treated with ipilimumab
Trial overview
KEYNOTE-522 was a randomized (2:1), double-blind, multicenter, placebo-controlled study evaluating the efficacy and safety of KEYTRUDA® (pembrolizumab) in combination with chemotherapy (carboplatin and paclitaxel followed by doxorubicin and cyclophosphamide [AC] or epirubicin and cyclophosphamide [EC]) given as a neoadjuvant treatment and continued as monotherapy adjuvant treatment after surgery, in newly diagnosed, previously untreated, high-risk early-stage TNBC patients (N=1,174)
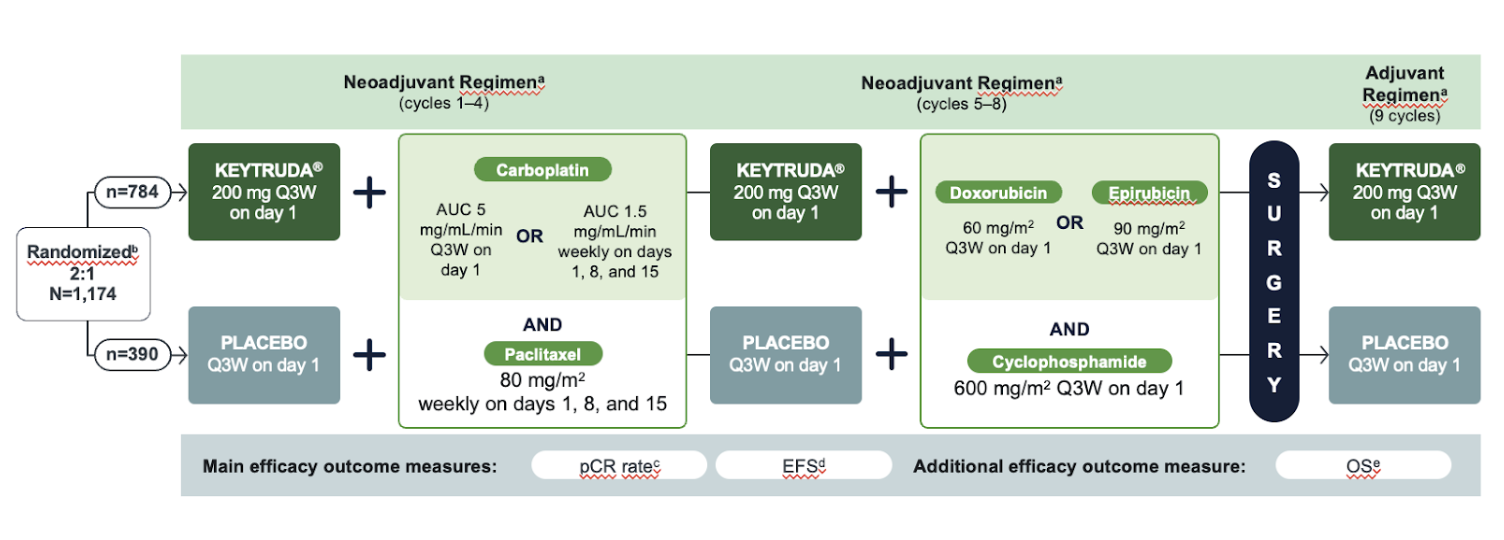
Eligibility Criteria
- Newly diagnosed, not previously treated high-risk, early-stage TNBC
- Tumor size >1 cm but ≤2 cm in diameter with nodal involvement or tumor size >2 cm in diameter regardless of nodal involvement
- Enrolled regardless of tumor PD-L1 expression
Primary Endpoints:
- Pathologic complete response (pCR), defined as absence of invasive cancer in the breast and lymph nodes (ypT0/Tis ypN0) and assessed by the blinded local pathologist at the time of definitive surgery
- Event-free survival (EFS), defined as the time from randomization to the first occurrence of any of the following events: progression of disease that precludes definitive surgery, local or distant recurrence, second primary malignancy, or death due to any cause.
Secondary Endpoints:
- Overall survival (OS)
KEYTRUDA® 2 mg/kg IV Q3W
(n=180)KEYTRUDA® 10 mg/kg IV Q3W
(n=181)Investigator’s choice of chemotherapy
(n=179)Regimens included:
– Dacarbazine 1000 mg/m2 intravenously every 3 weeks
– Temozolomide 200 mg/m2 orally once daily for 5 days every 28 days
– Carboplatin AUC 6 intravenously plus paclitaxel 225 mg/m2 intravenously every 3 weeks for four cycles then carboplatin AUC of 5 plus paclitaxel 175 mg/m2 every 3 weeks
– Paclitaxel 175 mg/m2 intravenously every 3 weeks
– Carboplatin AUC 5 or 6 intravenously every 3 weeksKEYTRUDA® 2 mg/kg or 10 mg/kg intravenously every 3 weeks are not recommended dosing regimens.
Primary endpoints
- Progression-free survival, as assessed by IRO review using RECIST 1.1
- Overall survival
Secondary endpoints
- Objective response rate
- Response duration
AE = adverse event; AUC=area under the curve; CI=confidence interval; ECOG PS=Eastern Cooperative Oncology Group Performance Status; FU=fluorouracil; HR=hazard ratio; IRO= independent radiology plus oncologist; OS=overall survival; PFS= progression-free survival; RECIST=response evaluation criteria in solid tumours.
*not statistically significant compared to multiplicity adjusted significance level of 0.01
a Based on final analysis
b Based on second interim analysis
† Based on the stratified Cox proportional hazard model
‡ p-Value based on stratified log rank test
d All patients as treatedOS & PFS data in patients with KEYTRUDA® vs investigator’s choice of chemotherapy
Overall Survival
Final AnalysisOSKEYTRUDA® 10 mg/kg Q3W vs chemotherapyHR=0.74† (95% CI: 0.57, 0.96; p=0.011*‡)aKEYTRUDA® 2 mg/kg Q3W vs chemotherapyHR=0.86† (95% CI: 0.67, 1.10; p=0.117*‡)aKEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
Kaplan-Meier Curve for overall survival in the KEYNOTE-002 trial (Intent-to-treat population)
Final Analysis
There was no statistically significant difference between pembrolizumab and chemotherapy in the final OS analysis that was not adjusted for the potentially confounding effects of crossover
Progression Free Survival
Second Interim AnalysisPFSKEYTRUDA® 10 mg/kg Q3W vs chemotherapyHR=0.50†(95% CI: 0.39, 0.64; p<0.0001‡)bKEYTRUDA® 2 mg/kg Q3W vs chemotherapyHR=0.57†(95% CI: 0.45, 0.73; p<0.0001‡)bKEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
Kaplan-Meier Curve for Progression-free Survival in the KEYNOTE-002 trial (Intent-to-treat population)
Second interim analysis
AE = adverse event; AUC=area under the curve; CI=confidence interval; ECOG PS=Eastern Cooperative Oncology Group Performance Status; FU=fluorouracil; HR=hazard ratio; IRO= independent radiology plus oncologist; OS=overall survival; PFS= progression-free survival; RECIST=response evaluation criteria in solid tumours.
*not statistically significant compared to multiplicity adjusted significance level of 0.01
a Based on final analysis
b Based on second interim analysis
† Based on the stratified Cox proportional hazard model
‡ p-Value based on stratified log rank test
d All patients as treatedKEYTRUDA® safety profile results in metastatic melanoma, in the KEYNOTE-002 trial
The most common grades 3-4 treatment-related adverse events were (treatment groups combined):
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionKEYTRUDA® 2 or 10 mg/kg
every 3 weeks
% (n=357)Chemotherapy
% (n=171)Fatigue0.84.7Vomiting0.62.4Diarrhea0.61.8Anemia0.35.3Nausea0.32.4Lymphocyte count decreased0.31.2KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
- Adverse reactions resulting in permanent discontinuation occurred in 12% of patients receiving KEYTRUDA®; the most common (≥1%) were general physical health deterioration (1%), asthenia (1%), dyspnea (1%), pneumonitis (1%) and generalized edema (1%).
- Adverse reactions leading to interruption of KEYTRUDA® occurred in 14% of patients.
Treatment-related adverse events, any grade (reported in ≥10% of patients) were:d
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionKEYTRUDA® 2 or 10 mg/kg
every 3 weeksChemotherapy
% (n=171)Fatigue25.836.3Pruritus22.13.5Rash10.94.7Decreased appetite7.015.2Nausea6.732.7Anemia3.420.5Vomiting3.415.2KEYTRUDA® 10 mg/kg Q2W or Q3W are not recommended dosing regimens.
For the complete list of treatment-related adverse events, please consult the Product Monograph
AE = adverse event; AUC=area under the curve; CI=confidence interval; ECOG PS=Eastern Cooperative Oncology Group Performance Status; FU=fluorouracil; HR=hazard ratio; IRO= independent radiology plus oncologist; OS=overall survival; PFS= progression-free survival; RECIST=response evaluation criteria in solid tumours.
*not statistically significant compared to multiplicity adjusted significance level of 0.01
a Based on final analysis
b Based on second interim analysis
† Based on the stratified Cox proportional hazard model
‡ p-Value based on stratified log rank test
d All patients as treated
References:
1. Merck Canada Inc. KEYTRUDA® Product Monograph. April 19, 2023.
2. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Melanoma Cancer, version 2. 2023. Available at: https://www.nccn.org/professionals/physician_gls/pdf/cutaneous_melanoma.pdf
CA-OOC-00027

