KEYTRUDA® for adjuvant treatment of RCC
-
Explore KEYTRUDA® as adjuvant treatment in the KEYNOTE-564 trial for adults with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions
KEYNOTE-564 trial: Placebo-controlled trial of KEYTRUDA® for the adjuvant treatment of adults with resected RCC
Trial overview
KEYNOTE-564 trial design
Multicentre, randomized, double-blind, placebo-controlled study in 994 patients with intermediate-high or high risk of recurrence, or M1 no evidence of disease (NED).
Intermediate-high risk category: pT2 with Grade 4 or sarcomatoid features; pT3, any grade without nodal involvement (N0) or distant metastases (M0).
High risk category: pT4, any grade N0 and M0; any pT, any grade with nodal involvement and M0.
M1 NED category: patients with metastatic disease who had undergone complete resection of primary and metastatic lesions.
- Patients must have undergone a partial nephroprotective or radical complete nephrectomy (and complete resection of solid, isolated, soft tissue metastatic lesion(s) in M1 NED participants) with negative surgical margins ≥4 weeks prior to the time of screening
- Patients with active autoimmune disease or a medical condition that required immunosuppression were ineligible
Imaging conducted Q12W for first two years, then Q16W from years 3 to 5, then Q24W annually.
Eligibility Criteria
- Histology confirmed clear cell renal cell carcinoma
- Intermediate-risk
- High risk
- M1 NED after surgery
- Surgery ≤12 weeks prior to randomization
- No prior systemic therapy
- ECOG PS 0 or 1
Treatment arms
KEYTRUDA® 200 mg Q3W
for up to 1 year until disease recurrence or unacceptable toxicity (n=496)
Placebo Q3W
for up to 1 year until disease recurrence or unacceptable toxicity (n=498)
Stratification Factors
- Metastatic status (M0 vs. M1 NED)
- M0 group further stratified:
- ECOG PS 0 vs. 1
- US vs. non-US
Primary endpoint
- Investigator-assessed disease-free survival (DFS) defined as time to recurrence, metastasis or death
Secondary endpoint
- Overall survival
Selected baseline demographics
- Baseline characteristics and demographics were generally comparable1,4
Adapted from Choueiri TK, et al.KEYTRUDA®
(n=496)Placebo
(n=498)Male70.0%72.1%Age (median [range])
>65 years60.0 (27-81)
31.9%60.0 (25-84)
34.5%ECOG PS=115.1%14.5%Geographic location
- North America
- European Union (incl. UK)
- Rest of the world
26.8%37.9%
35.3%
25.1%37.6%
37.3%Radical nephrectomy92.5%92.4%Sarcomatoid features
- Present
- Absent
- Unknown
10.5%84.1%5.4%
11.8%83.3%4.8%Disease risk category*
- M0, intermediate-high risk: pT2 with Grade 4 or sarcomatoid features: pT3 (any grade), N0 or M0
- M0, High-risk: pT4, any grade N0 and M0, any pT, any grade with nodal involvement and M0
- M1 NED: patient with metastatic disease who had undergone complete resection of primary and metastatic lesions
86.1%
8.1%
5.8%
86.9%
7.2%
5.8%PD-L1 combined positive score
- <1
- >1
- Missing data
25.0%73.6%1.4%
22.7%76.9%0.4%DFS=disease-free survival; ECOG= East Cooperative Oncology Group; M0=distant metastases; N0=nodal involvement; NED=no evidence of disease; PD-L1=programmed death ligand-1; pT=primary tumour; PS= Performance Status; Q3W=every 3 weeks; Q12W=every 12 weeks; Q16W=every 16 weeks; Q24W=every 24 weeks; RCC=renal cell carcinoma.
AE=adverse events; ALT=alanine aminotransferase; AST=aspartate aminotransferase; RCC=renal cell carcinoma; TRAEs=treatment-related adverse events.Intent to treat (ITT) population (interim analysis)
Disease-free Survival
A statistically significant improvement in DFS vs placebo demonstrated (primary endpoint)
32% improvement in instantaneous risk of the composite DFS (HR=0.68* [95% CI: 0.53, 0.87; p=0.0010†])
Kaplan-Meier Curve for Disease-Free Survival in the KEYNOTE-564 trial (Intent to Treat Population)
 * Based on the stratified Cox proportional hazard model.Endpoint (DFS)KEYTRUDA®
* Based on the stratified Cox proportional hazard model.Endpoint (DFS)KEYTRUDA®
200 mg Q3W
(n=496)Placebo
(n=498)Patients with an event22% (109/496)30% (151/498)Median in months (95% CI)NRNR24-month DFS rate (95% CI)77% (73, 81)68% (64, 72)
† Based on a stratified log-rank test. p-value is one-sided comparison with a boundary of 0.0114.Median follow-up time was 23.9 months (range: 2.5 to 41.5 months) at the prespecified interim analysis. The numbers at risk beyond the median follow-up of 24 months were small. Thus, the Kaplan–Meier estimates in the tails of the curves were based on censored data, were not stable, and should be interpreted with caution.
Overall Survival
At the time of the prespecified interim analysis, OS results were not yet mature with:1
18 deaths out of 496 patients
KEYTRUDA®
33 deaths out of 498 patients
PlaceboCI=confidence interval; DFS=disease-free survival; HR=hazard ratio; ITT=intent to treat; NR=not reached; OS=overall survival.
AE=adverse events; ALT=alanine aminotransferase; AST=aspartate aminotransferase; RCC=renal cell carcinoma; TRAEs=treatment-related adverse events.Demonstrated safety profile results of RCC patients treated with monotherapy KEYTRUDA® vs. placebo in the KEYNOTE-564 trial
The selected most common treatment-related adverse events reported in ≥10% of patients were:
Adapted from the KEYTRUDA® Product Monograph. The median duration of exposure to KEYTRUDA® was 11.1 months (number of administrations ranged 1 to 17)Adverse Reaction
KEYTRUDA®
(n=488)
%Placebo
(n=496)
%Any GradeGrade 3Grade 4Any GradeGrade 3Grade 4Fatigue20.3%0.8%014.3%00Pruritus18.6%0.2%011.5%00Hypothyroidism17.6%0.2%02.6%00Diarrhea15.8%1.6%010.3%00Rash15.0%0.8%07.3%00Hyperthyroidism10.2%0.2%0000Please refer to the KEYTRUDA® Product Monograph for complete safety information for the KEYNOTE-564 trials (TRAEs reported in ≥1% of patients).
- Serious TRAEs occurred in 12% of patients receiving KEYTRUDA®; the most common (≥1%) were adrenal insufficiency, colitis, and diabetic ketoacidosis
- Discontinuation of KEYTRUDA® due to TRAEs occurred in 17.6 % of patients; the most common (≥1%) were ALT increase (1.6%), colitis (1.0%), and adrenal insufficiency (1.0%)
- Dose interruption of KEYTRUDA® due to TRAEs occurred in 16.4% of patients; the most common (≥1%) were arthralgia (1.4%), diarrhea (1.4%), hypothyroidism (1.2%), fatigue (1.0%), ALT increase (1.0%), AST increase (1.2%)
AE=adverse events; ALT=alanine aminotransferase; AST=aspartate aminotransferase; RCC=renal cell carcinoma; TRAEs=treatment-related adverse events.
KEYTRUDA® in the first-line setting for advanced or metastatic RCC
-
EXPLORE KEYTRUDA® as combination therapy with axitinib in the KEYNOTE-426 trial for patients with advanced or metastatic RCC
KEYNOTE-426 trial: Open-label, active controlled trial of KEYTRUDA® + axitinib in treatment-naïve patients with advanced or metastatic RCC
Trial overview
- Randomized, multicenter, open-label, active-controlled trial
- Encompassed 861 patients with advanced or metastatic RCC with clear cell component, regardless of PD-L1 tumour status and IMDC risk group category (favourable, intermediate, or poor)
Treatment arms
KEYTRUDA® 200 mg Q3W
+ axitinib 5 mg BID orSunitinib 50 mg QD
Primary endpoints
- Overall survival
- Progression-free survival as assessed by BICR using RECIST 1.1
- Modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ
Secondary endpoints as assessed by BICR using RECIST 1.1
- Objective response rate
- Modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ
- Response duration
- Modified to follow a maximum of 10 target lesions and a maximum of 5 target lesions per organ
BICR=blinded independent central review; BID=twice a day; IMDC=International Metastatic RCC Database Consortium; PD-L1=programmed death ligand 1; QD=once daily; Q3W=every 3 weeks; RCC=renal cell carcinoma; RECIST=response evaluation criteria in solid tumours.
AE=adverse events; ALT=alanine aminotransferase; AST=aspartate aminotransferase; IMDC=International Metastatic RCC Database Consortium; KCRNC=Kidney Cancer Research Network of Canada; RCC=renal cell carcinoma.
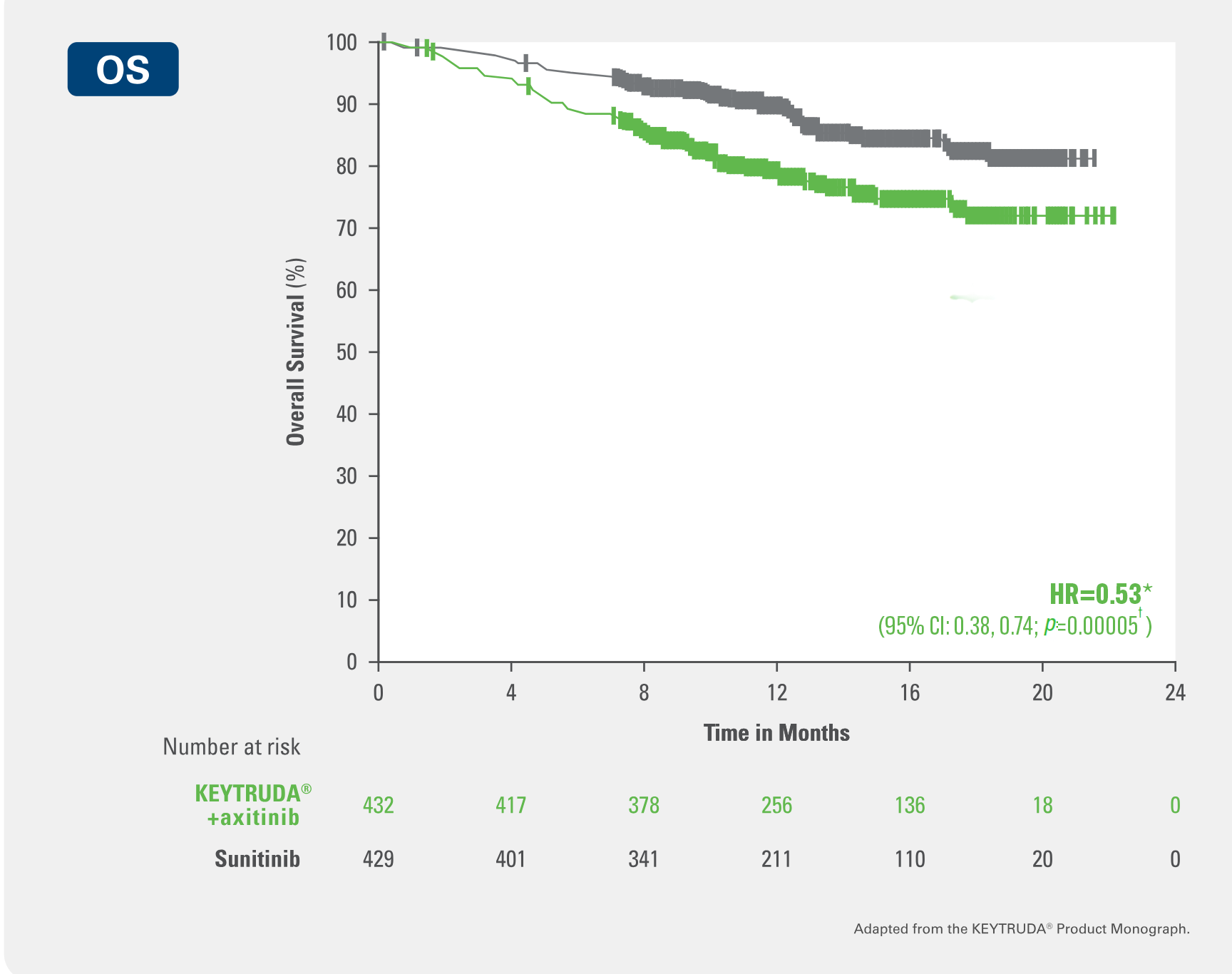
Intent to treat (ITT) population (interim analysis
Overall Survival endpoint (Primary endpoint)
Demonstrated improved overall survival for patients on KEYTRUDA® + axitinib vs sunitinib (primary endpoint)
Results of interim analysis in ITT demonstrated 47% reduction in the instantaneous risk of death with KEYTRUDA® + axitinib vs sunitinib (HR=0.53* [95% CI: 0.38, 0.74; p=0.00005†])
Kaplan-Meier Curve for Overall Survival in the KEYNOTE-426 trial (Intent to Treat Population)
Final OS analysis
The final OS analysis was performed at a median follow-up of 37.7 months.
median OSKEYTRUDA®+ axitinib Sunitinib193 patients with events225 patients with events45.7 months
(95% CI:43.6, NA)40.1 months
(95% CI: 34.3, 44,2)OSHR=0.73 (95% CI: 0.60, 0.88)Exploratory OS analysis
The updated analysis of OS in patients were:
- HR=1.17 (95% CI: 0.76, 1.80) in the group with IMDC favorable risk with KEYTRUDA® + axitinib vs sunitinib
- HR=0.67 (95% CI: 0.52, 0.86) in the group with IMDC intermediate risk with KEYTRUDA® + axitinib vs sunitinib
- HR=0.51 (95% CI: 0.32, 0.81) in the group with IMDC poor risk with KEYTRUDA® + axitinib vs sunitinib
Progression-Free Survival‡ (Primary endpoint)
31% reduction in the instantaneous risk of progression or death‡ with KEYTRUDA® + axitinib vs sunitinib (HR=0.69* [95% CI: 0.56, 0.84; p=0.00012†])
KEYTRUDA® + axitinib
(95% CI: 12.6, 17.7)Sunitinib (95% CI: 8.7, 12.5)15.1 monthsvs.11.1 monthsKaplan-Meier Curve for Progression-Free Survival in the KEYNOTE-426 trial (Intent to Treat Population)
Objective Response Rate‡ (secondary endpoint)
59% objective response rate§ with KEYTRUDA®+ axitinib (95% CI: 54, 64; p<0.0001¶) vs. sunitinib 36% (95% CI: 31, 40; p<0.0001¶; secondary endpoint)
KEYTRUDA® + axitinib
(n=432)vs.Sunitinib
(n=429)Complete response6%2%Partial response53%34%CI=confidence interval; HR=hazard ratio; IMDC=International Metastatic RCC Database Consortium; ITT=intent to treat; NA=not applicable; OS=overall survival.
* Based on the stratified Cox proportional hazard model.1
† Based on a stratified log-rank test.1
‡ The initial one-sided type 1 error rate levels for OS, PFS, ORR were 0.023, 0.002 and 0.025 respectively. The corresponding p-value bounds at the interim analysis for OS and PFS were 0.0001 and 0.0013, respectively. For ORR, the corresponding p-value bound after alpha reallocation from PFS and OS following pre-specified multiplicity adjustment was 0.025.1
§ Based on patients with a best overall response as confirmed complete or partial response.1
¶ Based on Miettinen and Nurminen method stratified by IMDC risk group and geographic region
AE=adverse events; ALT=alanine aminotransferase; AST=aspartate aminotransferase; IMDC=International Metastatic RCC Database Consortium; KCRNC=Kidney Cancer Research Network of Canada; RCC=renal cell carcinoma.
Demonstrated safety profile results of metastatic RCC patients treated with KEYTRUDA® in combination with axitinib vs. sunitinib in the KEYNOTE-426 trial
The most common Grades 3‒5 treatment-related adverse events reported for patients treated with either KEYTRUDA® + axitinib or sunitinib were:
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionKEYTRUDA® + axitinib
(n=429)
%Sunitinib
(n=425)
%Hypertension21.218.4Alanine aminotransferase increased12.12.6Diarrhea7.24.5Aspartate aminotransferase increased6.81.6Palmar-plantar erythrodysesthesia syndrome5.13.5- Treatment-related adverse events leading to interruption of KEYTRUDA® occurred in 41% of patients, regardless of action taken with axitinib
- The most common treatment-related adverse events leading to interruption of KEYTRUDA® (≥ 2%) were: ALT increased (9.1%); AST increased (8.4%); diarrhea (8.4%); and hyperthyroidism (2.1%)
- Fatal treatment-related adverse events occurred in 0.9% of patients receiving KEYTRUDA® in combination with axitinib
- These included 1 case each of myasthenia gravis, myocarditis, necrotising fasciitis, and pneumonitis
- Serious treatment-related adverse events occurred in 24% of patients receiving KEYTRUDA® in combination with axitinib
- Serious treatment-related adverse events in ≥ 1% of patients receiving KEYTRUDA® in combination with axitinib included: diarrhea (1.9%); ALT increased (1.4%); AST increased (1.2%); and pneumonitis (1.2%)
Selected treatment-related adverse events, any grade (reported in ≥10% of patients treated with either KEYTRUDA® + axitinib or sunitinib) were:
Adapted from the KEYTRUDA® Product Monograph.Adverse ReactionKEYTRUDA®+ axitinib
(n=429)
%Sunitinib
(n=425)
%Diarrhea4941.2Hypertension41.743.3Hypothyroidism31.528.0Fatigue30.333.4Palmar-plantar erythrodysesthesia syndrome27.739.5Alanine aminotransferase increased23.812.7Dysphonia22.82.8Aspartate aminotransferase increased22.613.9Decreased appetite21.924.9Nausea21.226.1Stomatitis14.220.2Mucosal inflammation12.821.2Dysgeusia9.330.4Thrombocytopenia1.922.1For the complete list of treatment-related adverse events, please consult the Product Monograph.
AE=adverse events; ALT=alanine aminotransferase; AST=aspartate aminotransferase; IMDC=International Metastatic RCC Database Consortium; KCRNC=Kidney Cancer Research Network of Canada; RCC=renal cell carcinoma.
References:
1. Merck Canada Inc. KEYTRUDA® Product Monograph. February 6, 2024.
2. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Kidney Cancer. Version 1.2024. © National Comprehensive Cancer Network, Inc. 2024. Available at: https://www.nccn.org/professionals/physician_gls/pdf/kidney.pdf.
3. Ljungberg B et al. EAU Guidelines on Renal Cell Carcinoma. © European Association of Urology 2023. March 2023. Available at: https://uroweb.org/guidelines/renal-cell-carcinoma.
4. Choueiri TK et al. Adjuvant pembrolizumab after nephrectomy in renal-cell carcinoma. NEJM. 2021;385: 683‒94.
5. Canil C et al. Management of advanced kidney cancer: Kidney Cancer Research Network of Canada (KCRNC) consensus update 2021. Can Urol Assoc J. 2021;15(4):84‒97.
CA-RCC-00170


