KEYTRUDA® with LENVIMA® for treatment of advanced or metastatic RCC
-
Explore KEYTRUDA® + LENVIMA® in the KEYNOTE-581/CLEAR trial for the treatment of adult patients with advanced or metastatic RCC with clear cell component who have not received prior systemic therapy for metastatic RCC
KEYNOTE-581/CLEAR trial: active-controlled trial in patients with advanced or metastatic RCC with clear cell component who have not received prior systemic therapy for metastatic RCC1
Trial overview
- Randomized, multicentre, open-label, active-controlled trial
- Encompassed 1,069 patients with advanced or metastatic RCC with clear cell component6
KEYNOTE-581/CLEAR STUDY:
 Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.
Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.Use of LENVIMA® in combination with everolimus for previously treated patients with RCC is off-label for adult patients with advanced (not amenable to curative surgery or radiation) or metastatic RCC with no prior systemic therapy for metastatic RCC. Administration of KEYTRUDA® + LENVIMA® beyond RECIST-defined disease progression is off-label; the recommended dosing for KEYTRUDA® + LENVIMA® states that treatment should be stopped when disease progression occurs.
KEYNOTE-581/CLEAR STUDY: Baseline characteristics were generally comparable among the 1,069 patients1,2
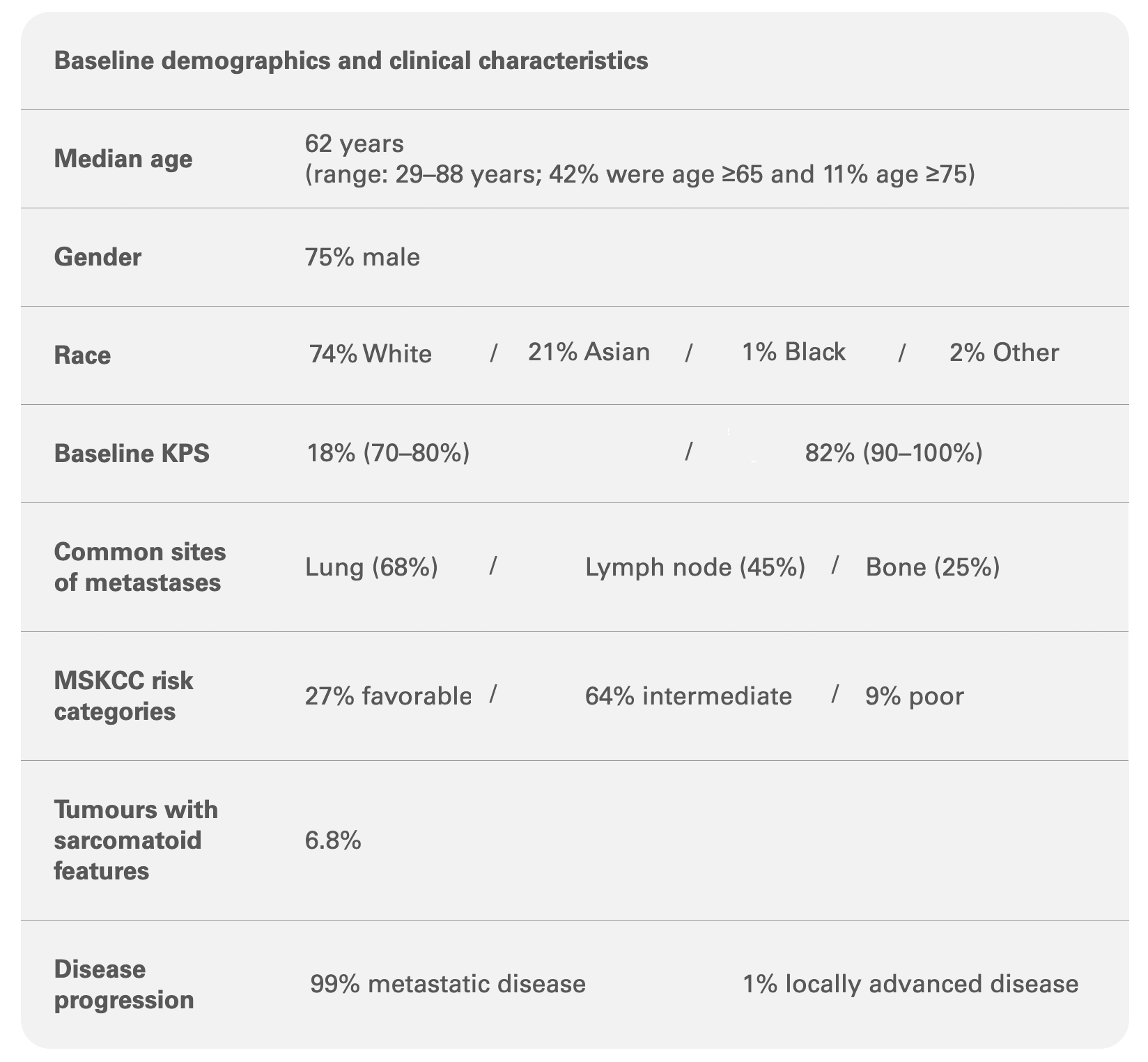 Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.1L=first-line; BICR=blinded independent central review; KPS=Karnofsky Performance Status; MSKCC=Memorial Sloan Kettering Cancer Center;
Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.1L=first-line; BICR=blinded independent central review; KPS=Karnofsky Performance Status; MSKCC=Memorial Sloan Kettering Cancer Center;
PD-L1=programmed death-ligand 1; Q3W=every three weeks; QD=once daily; RECIST=Response Evaluation Criteria in Solid Tumours.
* North America vs. Western Europe vs. “Rest of the World”.
† Favorable vs. intermediate vs. poor risk.§ Includes hypothyroidism, increased blood thyroid stimulating hormone, secondary hypothyroidism.1
¶ Includes diarrhea, gastroenteritis.1
** Includes aphthous ulcer, gingival pain, glossitis, glossodynia, mouth ulceration, mucosal inflammation, oral discomfort, oral mucosal blistering, oral pain, oropharyngeal pain, pharyngeal inflammation, stomatitis.1
†† Includes abdominal discomfort, abdominal pain, abdominal rigidity, abdominal tenderness, epigastric discomfort, lower abdominal pain, upper abdominal pain.1
‡‡ Includes asthenia, fatigue, lethargy, malaise.1
§§ Includes alanine aminotransferase increased, aspartate aminotransferase increased, blood bilirubin increased, drug-induced liver injury, hepatic enzyme increased, hepatic failure, hepatic function abnormal, hepatocellular injury, hepatotoxicity, hyperbilirubinemia, hypertransaminasemia, immune-mediated hepatitis, liver function test increased, liver injury, transaminases increased, gamma-glutamyltransferase increased.1
¶¶ Includes decreased appetite, early satiety.1
*** Includes arthralgia, arthritis, back pain, bone pain, breast pain, musculoskeletal chest pain, musculoskeletal discomfort, musculoskeletal pain, musculoskeletal stiffness, myalgia, neck pain, non-cardiac chest pain, pain in extremity, pain in jaw.1
††† Includes hemoglobinuria, nephrotic syndrome, proteinuria.1
‡‡‡ Includes acute kidney injury, azotemia, blood creatinine increased, creatinine renal clearance decreased, hypercreatininemia, renal failure, renal impairment, oliguria, glomerular filtration rate decreased, nephropathy toxic.1
§§§ Includes genital rash, infusion site rash, penile rash, perineal rash, rash, rash erythematous, rash macular, rash maculo-papular, rash papular, rash pruritic, rash pustular.1
¶¶¶ Includes palmar erythema, palmar-plantar erythrodysesthesia syndrome, plantar erythema.1
**** Includes essential hypertension, increased blood pressure, increased diastolic blood pressure, hypertension, hypertensive crisis, hypertensive retinopathy, labile blood pressure.1
†††† Includes all hemorrhage terms. Hemorrhage terms that occurred in 1 or more subjects in either treatment group include: anal hemorrhage, aneurysm ruptured, blood blister, blood loss anemia, blood urine present, catheter site hematoma, cerebral microhemorrhage, conjunctival hemorrhage, contusion, diarrhea hemorrhagic, disseminated intravascular coagulation, ecchymosis, epistaxis, eye hemorrhage, gastric hemorrhage, gastritis hemorrhagic, gingival bleeding, hemorrhage urinary tract, hemothorax, hematemesis, hematoma, hematochezia, hematuria, hemoptysis, hemorrhoidal hemorrhage, increased tendency to bruise, injection site hematoma, injection site hemorrhage, intra-abdominal hemorrhage, lower gastrointestinal hemorrhage, Mallory-Weiss syndrome, melaena, petechiae, rectal hemorrhage, renal hemorrhage, retroperitoneal hemorrhage, small intestinal hemorrhage, splinter hemorrhages, subcutaneous hematoma, subdural hematoma, subarachnoid hemorrhage, thrombotic thrombocytopenic purpura, tumour hemorrhage, traumatic hematoma, upper gastrointestinal hemorrhage.1KEYTRUDA® + LENVIMA® vs. sunitinib
Progression-free survival
KEYTRUDA® + LENVIMA® demonstrated significant improvements in progression-free survival (primary endpoint) vs. sunitinib in patients with advanced or metastatic RCC with no prior systemic therapy for RCC1,2
61% reduction in the risk of progression or death with KEYTRUDA® + LENVIMA® vs sunitinib (HR=0.39* [95% CI: 0.32, 0.49; p<0.0001†])
Kaplan-Meier curve for progression-free survival with KEYTRUDA® + LENVIMA® (n=355) vs. sunitinib (n=357)1,2
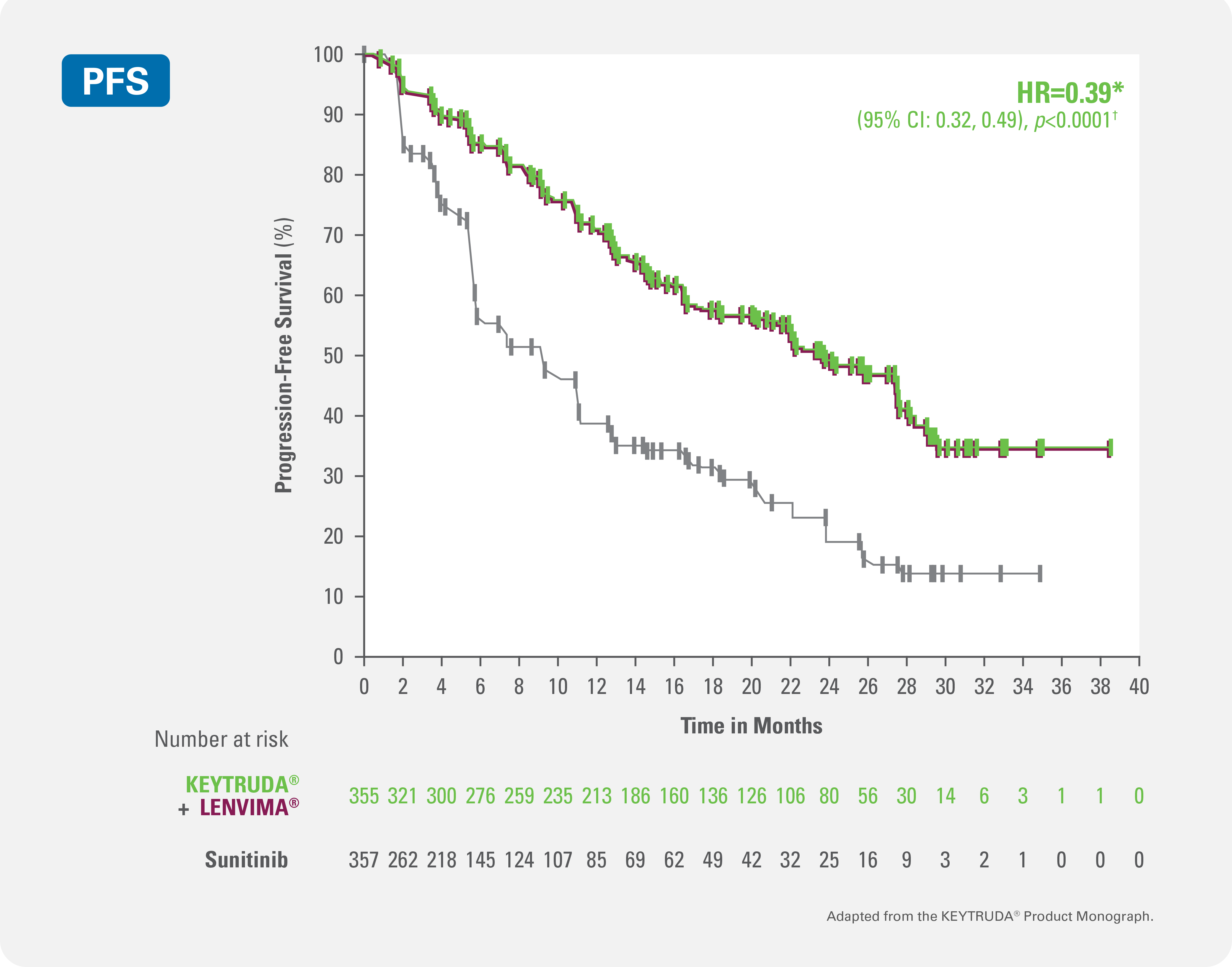 Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.
Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.KEYTRUDA® + LENVIMA®1,6
45%
(160/355 patients with event)Sunitinib1,6
57%
(205/357 patients with event)Median PFS was 23.9 months [95% CI: 20.8, 27.7] for KEYTRUDA® + LENVIMA®.
Median PFS was 9.2 months [95% CI: 6.0, 11.0] for sunitinib.1,2Overall Survival
KEYTRUDA® + LENVIMA® demonstrated significant improvements in overall survival (secondary endpoint) vs. sunitinib in patients with advanced or metastatic RCC with no prior systemic therapy for RCC1,2
34% reduction in the risk of death with KEYTRUDA® + LENVIMA® vs sunitinib (HR=0.66* [95% CI: 0.49, 0.88; p=0.0049†])
KEYTRUDA® + LENVIMA®
23%
(80/355 patients with event)Sunitinib
28%
(101/357 patients with event)Median overall survival follow-up time: 26.6 months (range: 0.03+, 46.13+ months).1,4
Median overall survival follow-up time was NR (33.6, NR) for KEYTRUDA® + LENVIMA® vs. NR (NR,NR) for sunitinib.1,2
The recommended dosing for KEYTRUDA® in adults is 200 mg q3w or 400 mg q6w until acceptable toxicity, disease progression or for up to 24 months.1
Kaplan-Meier Curve for Overall Survival in the KEYNOTE-581/CLEAR trial with KEYTRUDA® + LENVIMA® (n=355) vs. sunitinib (n=357)
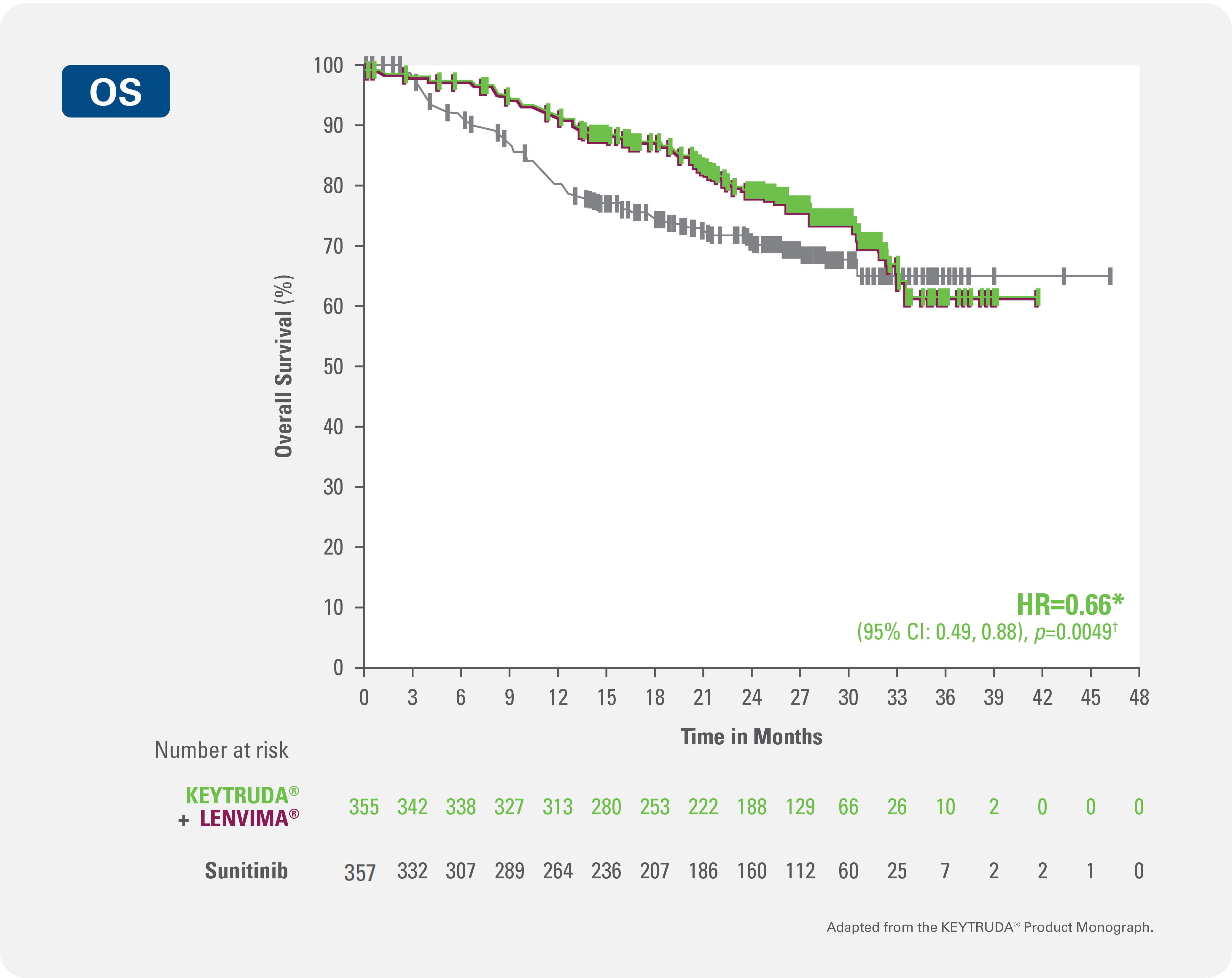 Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.
Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.Objective Response Rate
KEYTRUDA® + LENVIMA® demonstrated significant improvements in objective response rate (secondary endpoint) vs. sunitinib in patients with advanced or metastatic RCC with no prior systemic therapy for RCC1,2
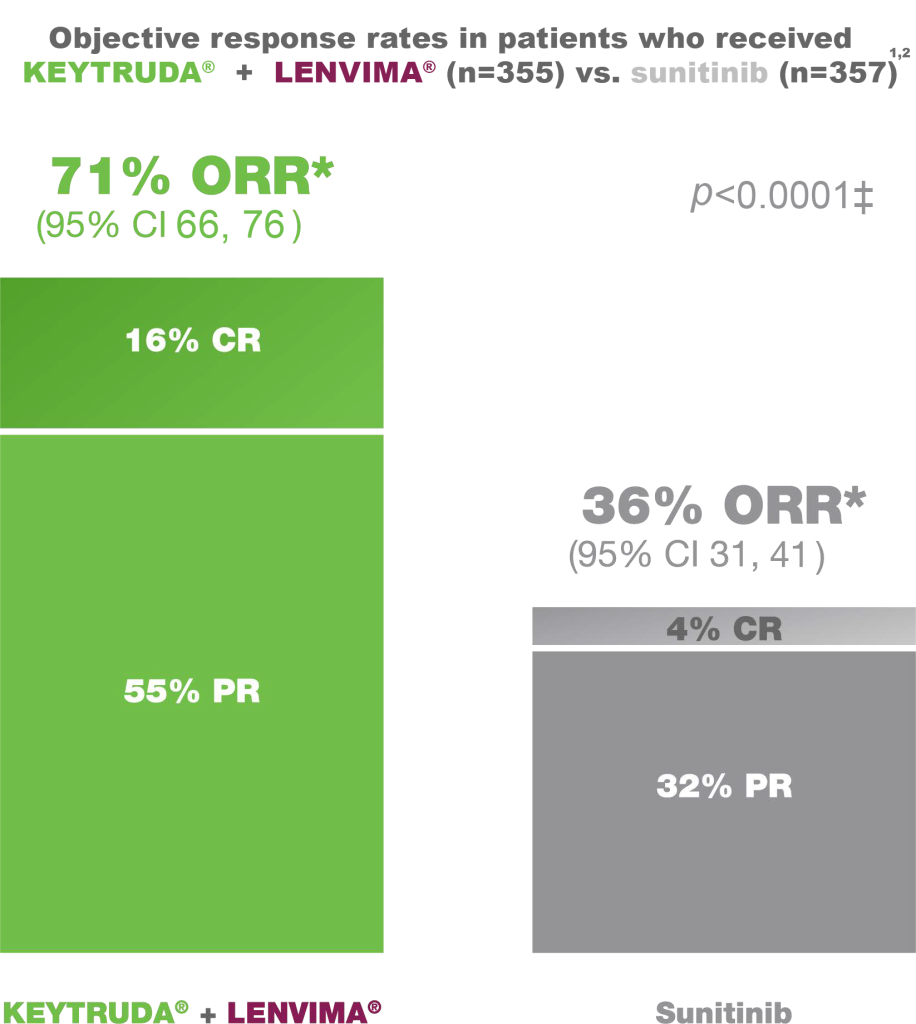 Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.
Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.KEYTRUDA® + LENVIMA® duration of response (exploratory endpoint) vs. sunitinib in patients with advanced or metastatic RCC 1,2
Suggested median duration of response for KEYTRUDA® + LENVIMA® vs. sunitinib in responders in KEYNOTE-581/CLEAR STUDY
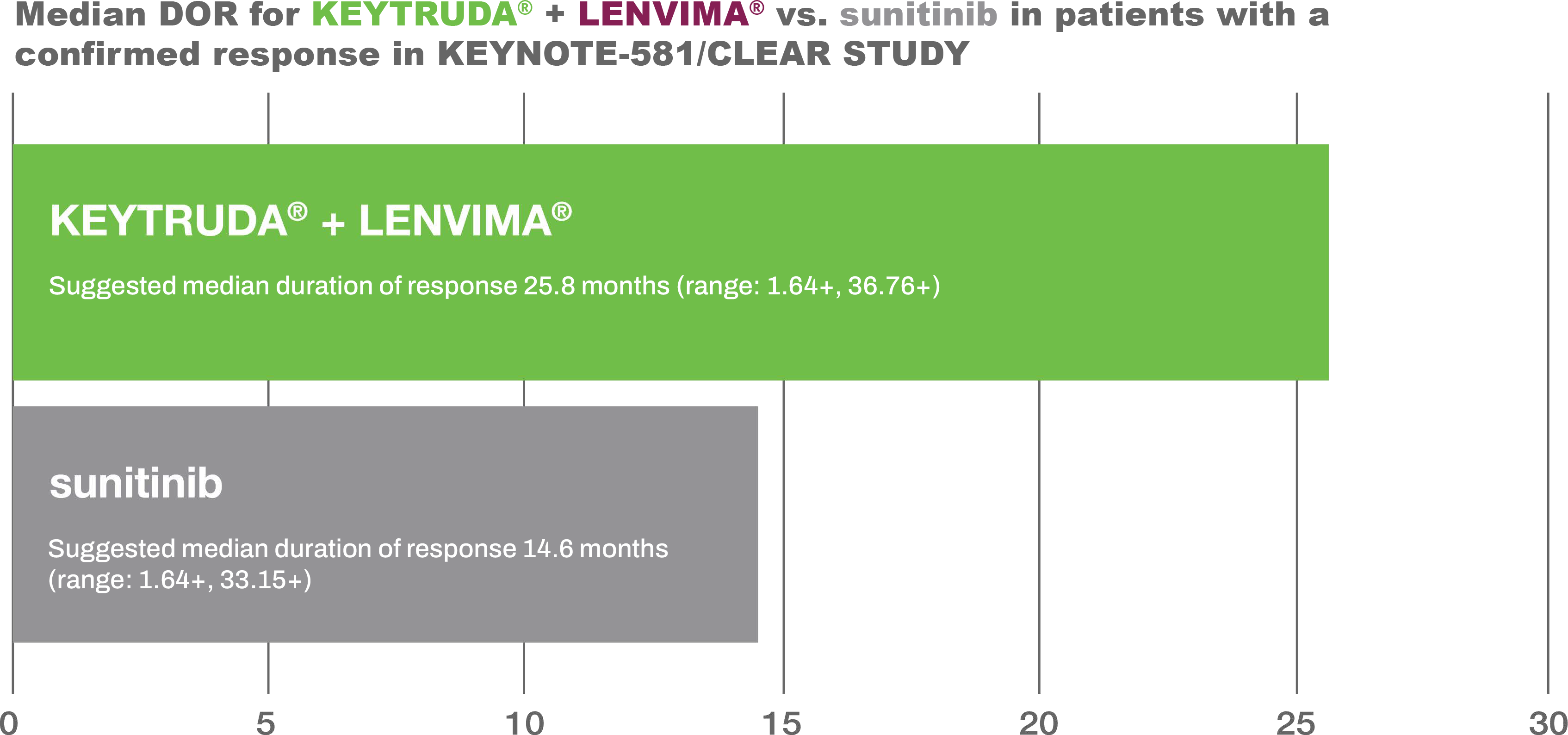 Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.
Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.
Tumour assessments were based on RECIST 1.1; only confirmed responses are included for ORR.CI=confidence interval; HR=hazard ratio; MSKCC=Memorial Sloan Kettering Cancer Center; q3w=every 3 weeks; q6w=every 6 weeks.
* Based on the stratified Cox proportional hazard model stratified by geographic region and MSKCC prognostic groups.
† Two-sided p-value based on stratified log-rank test, compared with a boundary of 0.0411 for PFS and 0.0161 for OS, respectively.
‡ Two-sided p-value based on Cochran-Mantel-Haenszel test.§ Includes hypothyroidism, increased blood thyroid stimulating hormone, secondary hypothyroidism.1
¶ Includes diarrhea, gastroenteritis.1
** Includes aphthous ulcer, gingival pain, glossitis, glossodynia, mouth ulceration, mucosal inflammation, oral discomfort, oral mucosal blistering, oral pain, oropharyngeal pain, pharyngeal inflammation, stomatitis.1
†† Includes abdominal discomfort, abdominal pain, abdominal rigidity, abdominal tenderness, epigastric discomfort, lower abdominal pain, upper abdominal pain.1
‡‡ Includes asthenia, fatigue, lethargy, malaise.1
§§ Includes alanine aminotransferase increased, aspartate aminotransferase increased, blood bilirubin increased, drug-induced liver injury, hepatic enzyme increased, hepatic failure, hepatic function abnormal, hepatocellular injury, hepatotoxicity, hyperbilirubinemia, hypertransaminasemia, immune-mediated hepatitis, liver function test increased, liver injury, transaminases increased, gamma-glutamyltransferase increased.1
¶¶ Includes decreased appetite, early satiety.1
*** Includes arthralgia, arthritis, back pain, bone pain, breast pain, musculoskeletal chest pain, musculoskeletal discomfort, musculoskeletal pain, musculoskeletal stiffness, myalgia, neck pain, non-cardiac chest pain, pain in extremity, pain in jaw.1
††† Includes hemoglobinuria, nephrotic syndrome, proteinuria.1
‡‡‡ Includes acute kidney injury, azotemia, blood creatinine increased, creatinine renal clearance decreased, hypercreatininemia, renal failure, renal impairment, oliguria, glomerular filtration rate decreased, nephropathy toxic.1
§§§ Includes genital rash, infusion site rash, penile rash, perineal rash, rash, rash erythematous, rash macular, rash maculo-papular, rash papular, rash pruritic, rash pustular.1
¶¶¶ Includes palmar erythema, palmar-plantar erythrodysesthesia syndrome, plantar erythema.1
**** Includes essential hypertension, increased blood pressure, increased diastolic blood pressure, hypertension, hypertensive crisis, hypertensive retinopathy, labile blood pressure.1
†††† Includes all hemorrhage terms. Hemorrhage terms that occurred in 1 or more subjects in either treatment group include: anal hemorrhage, aneurysm ruptured, blood blister, blood loss anemia, blood urine present, catheter site hematoma, cerebral microhemorrhage, conjunctival hemorrhage, contusion, diarrhea hemorrhagic, disseminated intravascular coagulation, ecchymosis, epistaxis, eye hemorrhage, gastric hemorrhage, gastritis hemorrhagic, gingival bleeding, hemorrhage urinary tract, hemothorax, hematemesis, hematoma, hematochezia, hematuria, hemoptysis, hemorrhoidal hemorrhage, increased tendency to bruise, injection site hematoma, injection site hemorrhage, intra-abdominal hemorrhage, lower gastrointestinal hemorrhage, Mallory-Weiss syndrome, melaena, petechiae, rectal hemorrhage, renal hemorrhage, retroperitoneal hemorrhage, small intestinal hemorrhage, splinter hemorrhages, subcutaneous hematoma, subdural hematoma, subarachnoid hemorrhage, thrombotic thrombocytopenic purpura, tumour hemorrhage, traumatic hematoma, upper gastrointestinal hemorrhage.1Demonstrated safety profile in patients with KEYTRUDA® in combination with LENVIMA®
Adverse events in ≥20% of patients with advanced or metastatic RCC
Adapted from the KEYTRUDA® and LENVIMA® Product Monographs.Adverse eventKEYTRUDA® + LENVIMA®
(N=352)Sunitinib
(N=340)All Grades
(%)Grades 3–4
(%)All Grades
(%)Grades 3–4
(%)Hypothyroidism§571320Diarrhea¶6210506Stomatitis**432432Nausea363331Abdominal pain††272181Vomiting263201Constipation251190Fatigue‡‡639568Hepatotoxicity§§259215Weight decreased30890.3Decreased appetite¶¶414311Musculoskeletal pain***584413Headache231161Proteinuria†††308133Acute kidney injury‡‡‡215162Dysphonia30040Rash§§§375171Palmar-plantar erythrodysesthesia syndrome¶¶¶294384Hypertension****56294320Hemorrhagic events††††275264- Treatment-related adverse events leading to permanent discontinuation of KEYTRUDA®, LENVIMA®, or both occurred in 37% of patients receiving KEYTRUDA® in combination with LENVIMA®; 29% of patients receiving KEYTRUDA® only, 26% of patients receiving LENVIMA® only, and 13% both.
- The most common adverse events (≥ 2%) leading to permanent discontinuation of KEYTRUDA®, LENVIMA®, or both were: pneumonitis (3%), myocardial infarction (3%), hepatotoxicity (3%), acute kidney injury (3%), rash (3%), and diarrhea (2%)
- Fatal treatment-related adverse events occurred in 4.3% of patients receiving KEYTRUDA® in combination with LENVIMA®
- These included cardio-respiratory arrest (0.9%), sepsis (0.9%), and one case (0.3%) each of arrhythmia, autoimmune hepatitis, dyspnea, hypertensive crisis, increased blood creatinine, multiple organ dysfunction syndrome, myasthenic syndrome, myocarditis, nephritis, pneumonitis, ruptured aneurysm, and subarachnoid hemorrhage
- Serious treatment-related adverse events occurred in 51% of patients receiving KEYTRUDA® in combination with LENVIMA®
- Serious adverse events in ≥ 2% of patients receiving KEYTRUDA® in combination with LENVIMA® included: hemorrhagic events (5%), diarrhea (4%), hypertension (3%), myocardial infarction (3%), pneumonitis (3%), vomiting (3%), acute kidney injury (2%), adrenal insufficiency (2%), dyspnea (2%), and pneumonia (2%)
Read more for guidance on KEYTRUDA® AE management.Go to AE management pageFor guidance on AE management in patients taking KEYTRUDA®+ LENVIMA®, download this handbook.
Help your patients taking KEYTRUDA®+ LENVIMA® keep track of their side effects by providing this personal diary.
§ Includes hypothyroidism, increased blood thyroid stimulating hormone, secondary hypothyroidism.1
¶ Includes diarrhea, gastroenteritis.1
** Includes aphthous ulcer, gingival pain, glossitis, glossodynia, mouth ulceration, mucosal inflammation, oral discomfort, oral mucosal blistering, oral pain, oropharyngeal pain, pharyngeal inflammation, stomatitis.1
†† Includes abdominal discomfort, abdominal pain, abdominal rigidity, abdominal tenderness, epigastric discomfort, lower abdominal pain, upper abdominal pain.1
‡‡ Includes asthenia, fatigue, lethargy, malaise.1
§§ Includes alanine aminotransferase increased, aspartate aminotransferase increased, blood bilirubin increased, drug-induced liver injury, hepatic enzyme increased, hepatic failure, hepatic function abnormal, hepatocellular injury, hepatotoxicity, hyperbilirubinemia, hypertransaminasemia, immune-mediated hepatitis, liver function test increased, liver injury, transaminases increased, gamma-glutamyltransferase increased.1
¶¶ Includes decreased appetite, early satiety.1
*** Includes arthralgia, arthritis, back pain, bone pain, breast pain, musculoskeletal chest pain, musculoskeletal discomfort, musculoskeletal pain, musculoskeletal stiffness, myalgia, neck pain, non-cardiac chest pain, pain in extremity, pain in jaw.1
††† Includes hemoglobinuria, nephrotic syndrome, proteinuria.1
‡‡‡ Includes acute kidney injury, azotemia, blood creatinine increased, creatinine renal clearance decreased, hypercreatininemia, renal failure, renal impairment, oliguria, glomerular filtration rate decreased, nephropathy toxic.1
§§§ Includes genital rash, infusion site rash, penile rash, perineal rash, rash, rash erythematous, rash macular, rash maculo-papular, rash papular, rash pruritic, rash pustular.1
¶¶¶ Includes palmar erythema, palmar-plantar erythrodysesthesia syndrome, plantar erythema.1
**** Includes essential hypertension, increased blood pressure, increased diastolic blood pressure, hypertension, hypertensive crisis, hypertensive retinopathy, labile blood pressure.1
†††† Includes all hemorrhage terms. Hemorrhage terms that occurred in 1 or more subjects in either treatment group include: anal hemorrhage, aneurysm ruptured, blood blister, blood loss anemia, blood urine present, catheter site hematoma, cerebral microhemorrhage, conjunctival hemorrhage, contusion, diarrhea hemorrhagic, disseminated intravascular coagulation, ecchymosis, epistaxis, eye hemorrhage, gastric hemorrhage, gastritis hemorrhagic, gingival bleeding, hemorrhage urinary tract, hemothorax, hematemesis, hematoma, hematochezia, hematuria, hemoptysis, hemorrhoidal hemorrhage, increased tendency to bruise, injection site hematoma, injection site hemorrhage, intra-abdominal hemorrhage, lower gastrointestinal hemorrhage, Mallory-Weiss syndrome, melaena, petechiae, rectal hemorrhage, renal hemorrhage, retroperitoneal hemorrhage, small intestinal hemorrhage, splinter hemorrhages, subcutaneous hematoma, subdural hematoma, subarachnoid hemorrhage, thrombotic thrombocytopenic purpura, tumour hemorrhage, traumatic hematoma, upper gastrointestinal hemorrhage.1
References:
- Merck Canada Inc. KEYTRUDA® Product Monograph. March 21, 2024.
- Eisai Limited. LENVIMA® Product Monograph. July 19, 2023.
- Canil C et al. Management of advanced kidney cancer: Kidney Cancer Research Network of Canada (KCRNC) consensus update 2021. Can Urol Assoc J. 2021;15(4):84‒97.
- Medical Council of Canada. Clinical laboratory tests: Adult normal values. Available at: https://mcc.ca/examinations-assessments/resources-to-help-with-exam-prep/normal-lab-values/. Last accessed April 19, 2023.
- NCCN Clinical Practice Guidelines in Oncology. Kidney Cancer, Version 2.2024.
- Motzer R et al. Lenvatinib plus Pembrolizumab or Everolimus for Advanced Renal Cell Carcinoma. N Engl J Med. 2021;384(14):1289-1300. doi: 10.1056/NEJMoa2035716
CA-KLH-00054

