KEYTRUDA® has 2 indications in TNBC, and an efficacy and safety profile demonstrated by 2 pivotal trials encompassing a total of 1449 patients. Learn more about when to consider KEYTRUDA® for your eligible TNBC patients1.
KEYTRUDA® is indicated for:
Treatment of adult patients with high-risk, early-stage, triple-negative breast cancer (TNBC) in combination with chemotherapy as neoadjuvant treatment, and then continued as monotherapy as adjuvant treatment after surgery1.
Treatment in combination with chemotherapy for adult patients with locally recurrent unresectable or metastatic triple-negative breast cancer (TNBC), who have not received prior chemotherapy for metastatic disease and whose tumours express PD-L1 (Combined Positive Score [CPS] ≥ 10) as determined by a validated test1.
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®†) for Breast Cancer2
Recurrent unresectable (local) or stage IV (M1) disease:
- Pembrolizumab + chemotherapy (albumin-bound paclitaxel, paclitaxel, or gemcitabine + carboplatin) is recommended as first-line therapy for PD-L1 CPS ≥ 10 triple-negative breast cancer.
High-Risk early-stage TNBC:
- Preoperative pembrolizumab + carboplatin + paclitaxel, followed by preoperative pembrolizumab + cyclophosphamide + doxorubicin or epirubicin, followed by adjuvant pembrolizumab as a preferred regimen.
Clinical Trials
KEYNOTE-355 (clinical study in locally recurrent unresectable or metastatic TNBC)
KEYNOTE-355KEYNOTE-355 Video
ViewKEYNOTE-522 (clinical study in high-risk, early-stage TNBC)
KEYNOTE-522KEYNOTE-522 Video
ViewBiomarker Testing
PD-L1 expression (CPS ≥ 10) is required for KEYTRUDA® treatment eligibility in locally recurrent unresectable or metastatic TNBC.
What is TNBC?
Triple-negative breast cancers (TNBC) are defined by the absence of hormone receptors ER and PR, and HER2.3 Biomarker testing for ER, PR, HER2 is needed to confirm TNBC status.3
Breast Cancer Staging per AJCC
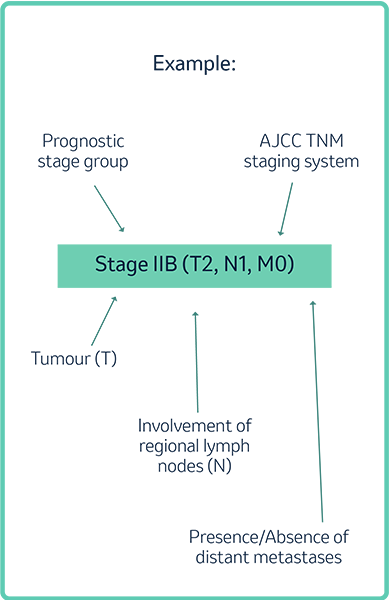
The most clinically useful staging system is the tumour, node, and metastasis (TNM) staging system, referred to as the American Joint Committee on Cancer (AJCC) TNM staging system.3
- It classifies cancers by the size and extent of the primary tumour (T), involvement of regional lymph nodes (N), and the presence or absence of distant metastases (M).3
- T, N, Stages I‒IV subgroups are denoted by capital letters according to cancer site stage grouping definitions and are used to expand the main groupings to provide more refined prognostic information.3
Anatomy of Breast Cancer
Anatomical Correlates of Breast Cancer Staging*3

*Description of anatomy related to staging has been simplified. For a full description of anatomy staging for breast cancer, including substages, see AJCC cancer staging manual.
In the latest edition of the American Joint Committee on Cancer (AJCC) staging manual, a major change in breast cancer staging involved incorporating the biomarker status (HER2, ER, and PR) as well as tumour grade in conjunction with anatomical T, M, N categories3
AJCC=American Joint Committee on Cancer, ER=estrogen receptor, HER2=human epidermal growth factor 2 receptor, NCCN=National Comprehensive Cancer Network®†, PR=progesterone receptor, TNBC=triple-negative breast cancer.
†All other trademarks are the property of their respective owner(s).
References:
1. KEYTRUDA® Product Monograph. Merck Canada Inc. February 6, 2024.
2. NCCN®. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Breast Cancer. March 23, 2023.
3. American Joint Committee on Cancer (AJCC). Cancer Staging Manual 8th edition. doi:10.1007/978-3-319-40618-3.
CA-OBR-00005
